Newsroom
View news releases and announcements distributed by EZ Newswire, the trusted source for business news.
Fires of Freedom Publishing Announces 'Fire, Brimstone and Blood' by Local Author Clay Ramsay
Fires of Freedom Publishing proudly announces the release of “Fire, Brimstone and Blood,” a new historical novel by local author Clay Ramsay. The book explores a pivotal yet often overlooked chapter of the American Revolutionary War: the brutal Southern Campaign in South Carolina.
While many of the war’s most famous battles were fought in the North, after four years of conflict the British had failed to subdue the rebellion there. In response, they launched a new strategy — holding their positions in the North while opening a Southern Campaign aimed at dividing and crippling the Patriot cause.
In South Carolina, under General Lord Cornwallis, the British seized major cities and ports and decimated organized Continental Army resistance. But in the state’s backcountry, they faced a different kind of war. Local volunteer militias — farmers and woodsmen, unpaid and untrained but fiercely determined — rose up to fight at a moment’s notice. Their efforts would change the course of the war in just a few months.
“Fire, Brimstone and Blood” tells their story through the voices of private militiamen, their wives and local commanders, bringing to life the courage and resilience of ordinary people whose sacrifices altered the nation’s history.
About Fires of Freedom Publishing
Fires of Freedom Publishing, LLC is the publishing home of author Clay Ramsay. Clay Ramsay graduated with honors and distinction from Washington and Lee University, earning degrees in English and classics. He was inducted into Phi Beta Kappa and later earned his MA in English at the University of Virginia.
Now enjoying semi-tranquil retirement on a farm with his wife, Mary, along with their dogs and horses, Ramsay draws inspiration from his lifelong passion for American history and the revolutionary ideals of the Founding Fathers. Encouraged for decades by his mother to write a book, he finally answered the call — crossing one off the bucket list with “Fire, Brimstone and Blood.”
For more information, visit firesoffreedom.my.canva.site.
Media Contact
Clay Ramsay
Fires of Freedom Publishing
cowboytuff76@gmail.com
+1 434-996-2817



Unlisted Expands Network of Real Estate Professionals with Bob Miller of RE/MAX Premier Choice
Unlisted, a groundbreaking digital real estate community focused on the 98% of homes that aren’t for sale—yet, today announced that Bob Miller of RE/MAX Premier Choice has joined the platform as a Local Expert for buyers and homeowners in Dublin, Ohio, representing the 43016 and 43017 ZIP codes.
Unlisted’s technology is designed to open up possibilities for everyone in the real estate ecosystem: buyers, homeowners, and agents alike. Buyers gain new ways to discover homes they love beyond what’s listed on the MLS. Homeowners gain tools to better understand interest in their property and connect with potential buyers. Real estate professionals gain a program that elevates their expertise and brings more possibilities into view for their clients.
Unlisted uses publicly available data to create a digital property profile for every home in the country. These records can be quickly searched and organized into curated lists of homes that meet a buyer’s criteria. Though not for sale, buyers can join a Waitlist for their favorite homes, creating new possibilities for buyers and agents to connect and giving homeowners options they may not have considered.
Each vetted real estate professional that partners with Unlisted receives an agent profile linked to every home in their ZIP code. Only one agent is selected per ZIP, highlighting their expertise and credibility in that market. As Unlisted continues to expand this network nationwide, the goal remains the same: to provide buyers, homeowners, and agents with a platform built on connection, expertise, and meaningful insights.
Bob Miller brings decades of real estate experience to his role as Unlisted’s local expert in Dublin. Having built a career on trusted relationships and community knowledge, Miller is also embracing innovation by adopting new tools that reflect the changing landscape of real estate. His decision to join Unlisted demonstrates how the platform empowers agents of all backgrounds—whether seasoned professionals with years of traditional experience or younger agents leading with tech-savvy practices.
“Real estate never stands still,” says Miller. “Even after years in this business, I’m excited to keep learning and keep evolving. Partnering with Unlisted allows me to offer my clients in Dublin the best of both worlds: the experience of a trusted advisor and the advantages of new technology.”
“We’re so glad to welcome Bob Miller as our Local Expert in the 43016 and 43017 ZIP codes,” said Katie Hill, founder and CEO of Unlisted. “Bob represents the best of both tradition and innovation, which is exactly what makes him such an asset to homeowners and buyers in Dublin. We look forward to the opportunities his expertise and dedication will unlock for the community.”
To learn more about Unlisted, visit UnlistedHomes.com. For Unlisted for Agents, visit UnlistedHomes.com/Agents.
To learn more about Bob Miller, visit his Unlisted Profile, Miller's website, or RE/MAX.
About Unlisted
Unlisted focuses on the 98% of homes that aren’t for sale—yet. The company empowers home buyers to join the Waitlist for homes they love that aren’t for sale—in other words, unlisted. At the same time, homeowners collect a Waitlist of interested buyers for whenever the time comes to sell, giving everyone a head start. Unlisted also allows homeowners to control how their home is presented online; they can create a stunning up-to-date property profile that shows off the home’s best features and attracts more interest. With more time and more connection, Unlisted is a more human way to explore real estate that drives better outcomes for all. For more information, visit UnlistedHomes.com.
Media Contact
Sophia Jacomet
sophia@unlistedinc.com



Paris Peace Symposium Highlights Shaykh Mohamed Faouzi al-Karkari's Global Efforts
In celebration of the International Day of Peace, Paris hosted the Rotary Symposium for Peace, a distinguished gathering that united leaders from diverse spiritual, cultural, and political backgrounds. Central to this event was Shaykh Mohamed Faouzi al-Karkari, founder of the Al-Karkari Institute and a prominent figure in contemporary Islamic spirituality.
Representing the principles of Sufism, Shaykh al-Karkari participated in a pivotal panel discussion on secularism, moderated by Claire Derville of France 2's "Le Jour du Seigneur." Alongside leaders from Christianity, Hinduism, and French civic society, he illuminated the nuanced perspectives on secularism between Morocco and France, advocating for dialogue, mutual understanding, and respect.
Shaykh al-Karkari drew from the Qur’an to emphasize humanity’s shared purpose and the value of diversity, citing:
“We created you as nations and tribes so that you may know one another.” (Qur’an, Surah 49:13)
Known internationally for his vision of spiritual unity and peace, Shaykh al-Karkari has inspired more than two million followers through the Karkariya order and Al-Karkari Institute. The institute is renowned for its commitment to promoting interfaith harmony and spiritual education across continents.
At the close of the symposium, Shaykh al-Karkari was formally recognized for his tireless dedication to peace-building and fostering connections among diverse peoples. This honor reflects his ongoing mission to bridge communities and cultivate a spirit of compassion and brotherhood worldwide.
About Al-Karkari Institute
The Al-Karkari Institute aims to engage in research and discourse on the diverse dimensions of Sufism. By exploring the historical significance and contemporary relevance of tasawwuf, we aim to illuminate the path of Islamic inner transformation for a global audience of seekers. For more information, visit www.karkari.org.
Media Contact
Alan Noble
alan@karkari.org



WuXi AppTec Marks 25 Years as a Trusted Partner in Global Drug Discovery, Development, and Manufacturing
WuXi AppTec marks its 25th anniversary this year as a trusted partner to innovators worldwide. Over the past quarter century, the company’s integrated platform has reduced drug development timelines, helping advance breakthrough medicines for patients in the U.S. and around the globe.
Proven Global Partner Through Measurable Results
Since 2000, WuXi AppTec has provided integrated R&D and manufacturing solutions to innovators across the spectrum — from early-stage biotech to the world’s largest pharmaceutical companies. Its multi-jurisdictional regulatory expertise spans FDA, EMA, and NMPA guidelines, enabling faster, compliant market access for therapies around the world.
WuXi AppTec has contributed to reducing barriers to drug discovery, development, and manufacturing by investing in advanced technologies, expanding multi-region manufacturing capacity.
Integrated CRDMO Platform
WuXi AppTec’s Contract Research, Development, and Manufacturing Organization (CRDMO) model supports nearly 6,000 partners across 30+ countries through a distributed, resilient network of facilities. This integrated approach ensures that every stage — from early discovery to large-scale commercial manufacturing — advances seamlessly with efficiency, confidence, and world-class quality, backed by ISO/IEC 27001-certified information security management systems, customer-controlled access protocols, and annual third-party security audits.
With operations across Asia, Europe, and North America, the company meets the highest global regulatory standards — including zero critical FDA observations in 2024 — and engages proactively with health authorities worldwide.
Shaping the Future of Drug Modalities
In 2025, WuXi AppTec continued to expand its TIDES (oligonucleotide and peptide) manufacturing capacity, with its total reactor volume of Solid Phase Peptide Synthesizers expected to exceed 100,000L by end of 2025. These investments are helping accelerate the development of next-generation therapies in cancer, rare diseases, and other areas of urgent unmet medical need.
Commitment to Global Health and Sustainability
WuXi AppTec has achieved carbon neutrality across discovery operations and is committed to net-zero emissions by 2030. Compared to its 2020 baseline, the company has reduced water usage intensity by over 36% last year — reflecting its commitment to advancing science, enabling broader patient access, and driving measurable social impact worldwide.
Looking Forward
Building on 25 years as a trusted partner, WuXi AppTec will continue to advance breakthrough medicines through measurable improvements in speed, quality, and patient access — while maintaining the highest security and regulatory standards to serve patients worldwide.
Learn more at wuxiapptec.com.
For media inquiries, contact mediainquiries@wuxiapptec.com.



The Fitness Revolution: How RITFIT Is Leading on Accessibility and Inclusion
This year, as RITFIT celebrates its 10th anniversary, the brand is reaffirming its belief that fitness should be accessible to all. In an industry often dominated by aesthetic goals and performance metrics, RITFIT is reshaping the conversation and challenging how people think about fitness.
Pushing beyond the predictable world of gym culture, RITFIT has launched a standout initiative: its monthly Disability Support Program, which provides people with disabilities access to professional-grade home fitness gear and the motivation they need to achieve their goals.
Inclusive From the Ground Up
Accessibility isn’t an afterthought — it’s a foundation. RITFIT believes fitness should be available to everyone, which is why its full range of home and commercial gym equipment, from resistance bands to power cages, is designed for versatility, durability, and affordability.
But beyond innovative equipment, it’s the company’s social mission that sets it apart. Through consistent outreach and community partnerships, RITFIT provides free or subsidized training equipment to people with disabilities. By removing financial and logistical barriers, the brand ensures everyone can access a safe, empowering, and sustainable fitness routine — right from their own living rooms.
Kendra Herber: A Life Built on Strength
For Kendra Herber, fitness is more than a routine — it’s her anchor. Born with a limb difference, she has spent much of her life challenging assumptions about ability, femininity, and strength. Today, she’s a speaker, author, and advocate, as well as a proud RITFIT user and role model in the adaptive fitness community.
With RITFIT’s reliable, adjustable equipment tailored to her training style, Kendra stays active at home while caring for her daughters and pursuing her passion for advocacy, inspiring others with her message of resilience.
Kevin Ortiz: Representation Matters
As a wheelchair user and adaptive fitness coach, Kevin Ortiz has built his platform on motivation, representation, and authenticity. His Instagram (@wheelchairkev) showcases pull-ups, push-ups, and an unstoppable drive that leaps off the screen.
Through RITFIT’s Disability Support Program, Kevin received gear that elevated his workouts and training routines. Even more importantly, he found a brand that shares his values. Today, Kevin stands as one of RITFIT’s most visible ambassadors, using his platform to promote accessibility and guide people with disabilities who want to deepen their fitness journey.
Designing for Real Life, Not Just the Gym
The RITFIT brand is not just for elite athletes or influencers. They pride themselves on catering to all experience levels and body types by offering equipment designed to fit real homes, real lives, and real goals. Additionally, their equipment is all rigorously tested and trusted by everyday users, personal trainers, and rehab specialists alike.
With a product line ranging from squat racks and wall-mounted storage to adjustable benches and resistance bands, RITFIT has always prioritized user-first design. That means focusing on comfort, adaptability, and functionality — not just looks.
Building More Than Muscle
What started as a gym equipment company is quickly becoming a community, and every month, RITFIT spotlights new stories of people from across the country who are using fitness to heal, connect, and grow. Their website and social media channels are filled with transformation stories, adaptive workout routines, and reminders that strength comes in many forms.
Ready to Get Involved?
The brand invites users not just to purchase equipment, but to join a mission. If you are a fitness professional, a rehab specialist, or someone simply looking for support on your journey, RITFIT makes it easy to step in and step up:
- Visit www.ritfitsports.com/pages/disability-program to learn more about the Disability Support Program
- Nominate an individual or organization for support
- Donate, partner, or sponsor adaptive equipment
- Follow @ritfitsports and join a growing community of inclusive fitness advocates
A Future with No Limits
RITFITis offering an industry long dominated by aesthetics and exclusivity, a fresh look at a world where everybody belongs. It is reshaping fitness norms, lifting voices, and celebrating what’s possible when we invest in people, not just products. With stories like Kendra’s and Kevin’s leave no doubt that the future of fitness is not only stronger — it’s more inclusive, more empowering, and more human.
About RITFIT
Founded in 2015, RITFIT — a name derived from “Right” and “Fitness” — empowers individuals to take control of their health through premium, accessible and user-friendly home gym equipment. With more than 30 patents and over 200,000 customers worldwide, RITFIT is redefining home fitness by making strength training simple, strong and effective. To learn more, visit www.ritfitsports.com.
Media Contact
RITFIT Marketing
marketing@ritfitsports.com



American Herbal Pharmacopoeia Publishes Kava Rhizome and Root Monograph, Marking a Watershed Moment for Informed Policy and Safe Use of Noble Kava
American Herbal Pharmacopoeia® (AHP) has released its long-anticipated kava rhizome and root monograph, marking a watershed moment in the global dialogue on noble kava’s role as a safe, culturally significant, and pro-social beverage. This comprehensive scientific and ethnobotanical review provides regulators, industry leaders, and consumers with a clear framework for understanding both the safety and the benefits of noble kava.
The monograph’s conclusion underscores what Pacific Islanders have known for centuries: when used appropriately and responsibly, noble kava is safe. The document affirms that the most common adverse effect from high-volume, long-term use is kava dermopathy (kani kani), a reversible and rare skin condition. Importantly, large-scale reviews demonstrate that concerns about hepatotoxicity, which led to near-global restrictions two decades ago, are exceedingly rare, often idiosyncratic, and associated with the use of organic solvent extracts and non-noble kava use. Both traditionally prepared noble kava and safe, food-grade CO₂ extractions have consistently shown strong safety profiles when consumed as intended.
The AHP review consolidates decades of clinical trial data and traditional knowledge, showing that noble kava compares favorably to many commonly used foods and dietary supplements when consumed responsibly. Research consistently finds that adverse effects are rare, mild, and often indistinguishable from placebo, reinforcing noble kava’s status as a safe, pro-social beverage when consumed responsibly.
“This monograph is a turning point,” said Douglas La Rose, Executive Director of the Kava Coalition. “It brings together decades of clinical evidence, traditional knowledge, and modern science to affirm the safety and cultural importance of noble kava. For policymakers, it offers a roadmap to craft regulations that ensure global access to noble kava products, safeguard legal access for Pacific Island diaspora communities, and validate noble kava as a safe food that consumers worldwide should be able to enjoy.”
The release of the AHP kava rhizome and root monograph is expected to serve as a cornerstone reference for governments, public health authorities, and industry stakeholders worldwide as they seek balanced, evidence-based approaches to regulating and promoting noble kava.
About Kava Coalition
The Kava Coalition is an alliance of noble kava consumers, experts, and industry leaders committed to advancing kava education, advocacy, and choice. By promoting responsible consumption, market transparency, and cultural respect, the Coalition works to ensure that noble kava is widely recognized, accessible, and valued as a safe, pro-social beverage. For more information, visit www.kavacoalition.org.
Media Contact
Patrick George
info@globalkratomcoalition.org
+1 916-202-1982



StartupMafia Unveils 10 Essential Digital Solutions Driving Business Growth in 2025
Businesses today face growing challenges that require integrated digital tools. To help companies navigate this landscape, StartupMafia has identified ten essential platforms for sustainable growth in 2025.
These solutions span analytics, AI, localization, social media, software development, and employee well-being — offering enterprises a toolkit to stay competitive in fast-changing markets.
Lucimed – Innovators in Light Therapy
Lucimed, a Belgian health-tech company, specializes in light therapy designed to improve workplace well-being. Its flagship product, the Luminette®, delivers clinically proven benefits for combating seasonal affective disorder, regulating sleep cycles, and boosting daily energy levels.
Trusted by over 300,000 users worldwide, Luminette has become a go-to solution for organizations aiming to enhance productivity through employee health. By offering portability and medical-grade efficiency, Lucimed provides businesses with a simple yet powerful way to support staff performance.
Magic Data Recovery – Reliable File Restoration
Magic Data Recovery addresses one of the most critical risks of the digital era: data loss. The platform enables users to recover deleted, formatted, or inaccessible files from hard drives, USBs, and memory cards with high success rates.
Its intuitive interface and dual scanning modes — quick and deep — make it suitable for both IT professionals and individual users. In a business environment where data security and continuity are paramount, Magic Data Recovery stands out as a dependable recovery solution.
Instant Famous – Social Media Growth Services
Instant Famous provides businesses and creators with tools to boost visibility on platforms like Instagram, TikTok, YouTube, and Facebook. The service focuses on delivering authentic engagement — followers, likes, comments, and views — to strengthen social credibility.
With fast delivery, secure payment systems, and a strong track record of delivering promised results, Instant Famous has established itself as a trusted option for companies seeking rapid audience growth without compromising safety or efficiency.
Crowdin – AI-Driven Localization Platform
Crowdin is a leading localization software and translation management solution helping companies scale their products and content for global markets. With over 700 integrations, the platform centralizes translation workflows and enables continuous localization.
Crowdin leverages advanced AI localization and in-context review tools to deliver highly accurate, cost-effective translations at scale. With advanced security features and ISO 27001 certification, it's a trusted solution for enterprises looking to expand globally without compromising on security or brand consistency.
AIParabellum – Curated AI Tool Directory
AIParabellum simplifies the adoption of artificial intelligence by curating hundreds of AI tools across industries and use cases. Each listing provides key features, pricing details, and direct access links, allowing businesses to quickly identify and compare the most relevant solutions.
For marketing, development, and research teams, AIParabellum serves as a one-stop hub for discovering practical AI applications. Its structured approach helps companies stay ahead of technology trends and integrate AI more strategically.
Site.pro – Unified ERP and Website Builder
Site.pro offers an all-in-one ERP platform combining accounting tools, email services, and an AI-driven website builder. The integrated system allows businesses to manage financial operations, e-commerce, and online presence within a single interface.
In September 2025, Site.pro launched its live AI website builder, enabling users to generate prototypes and refine them via chatbot assistance. This combination of ERP and AI makes Site.pro especially attractive to small and medium-sized enterprises seeking efficiency.
Seedium – AI-Enhanced Software Development
Seedium is a global web and mobile development company leveraging AI to accelerate product design and delivery. By combining automation with proven engineering practices, Seedium helps businesses scale digital solutions faster and more effectively.
With over 200 completed projects across industries such as healthcare, fintech, real estate, and e-commerce, the company has earned a reputation as a reliable development partner. Its AI-powered approach ensures smarter workflows and reduced time-to-market.
Similarweb – Competitive Market Intelligence
Similarweb provides businesses with digital insights through advanced web traffic analytics. The platform tracks market trends, customer behavior, and competitor performance, giving organizations the intelligence needed to refine strategies and capture opportunities.
Widely used by enterprises, investors, and analysts, Similarweb enables data-driven decisions across marketing, product development, and growth planning. Its combination of accuracy and scale makes it an industry standard for competitive research.
Originality.AI – AI Content Verification
Originality.AI offers businesses and publishers a critical safeguard against AI-generated plagiarism. The platform scans written content for originality, authorship, and compliance with editorial standards.
As companies increasingly rely on automated writing tools, Originality.AI helps ensure quality, authenticity, and brand integrity. Its focus on accuracy and transparency has positioned it as a trusted partner for media outlets, universities, and corporate communication teams.
MobiDev – Trusted Software Development Partner
MobiDev is a software engineering company helping businesses design, build, and modernize digital products. Since 2009, it has supported companies in scaling operations through web, mobile, and AI-powered solutions tailored to industry needs.
The firm focuses on long-term collaboration and product sustainability, assisting clients in adapting to market changes and staying ahead of technology trends. MobiDev provides practical expertise that enables organizations to launch future-ready solutions and maintain their competitive edge.
From health and productivity to data recovery, AI, and market intelligence, these ten solutions highlight the evolving priorities of businesses in 2025. Each platform provides a practical tool to address modern challenges — ensuring companies can scale efficiently, expand globally, and remain competitive in a technology-driven economy.
About StartupMafia
StartupMafia is an international platform that tracks and highlights the most promising companies, services, and innovations shaping the digital economy. By spotlighting trusted players across industries, StartupMafia helps entrepreneurs, investors, and brands discover reliable partners and growth opportunities. For more information, visit startupmafia.eu.
Media Contact
StartupMafia Editorial Team
team@startupmafia.eu
.jpeg)
.jpeg)

Powering the AI Revolution — 3M’s Advanced Solutions Help Drive Efficiency, Speed, and Reliability in Data Centers
As artificial intelligence (AI) workloads surge, U.S. data centers are under immense pressure to grow capacity while minimizing energy demand. A recent analysis from Epoch AI highlights that computational performance per watt in AI supercomputers has increased by 34% every year from 2019 to 2025, driven by innovations in cooling, power delivery, and energy efficient chip design. To sustain this momentum, data centers must optimize every aspect of power and thermal management.
According to a July 2025 report by 174 Power Global, AI workloads could push U.S. data center electricity demands sharply upward — AI alone may soon account for nearly eight percent of national power use and global demand may double by 2030. Flexible, smart energy architectures are vital to meeting this challenge, aligning with national goals to lead the AI era responsibly.
“From the grid to the rack, 3M innovations are designed to help hyperscalers and data center operators build faster, operate more efficiently and adapt to new workloads with confidence,” said Alex An, vice president, 3M Data Center Vertical. “Whether it’s enabling high-bandwidth, low-latency optical interconnects, solving mission-critical thermal challenges, improving the efficiency of power delivery, or improving hot aisle containment, 3M is helping data centers meet their increasing challenges of AI-driven growth by pushing the limits of our materials science, minimizing energy footprints and helping increasing connectivity speeds.”
3M innovations: Efficiency, performance, and sustainability
The 3M Cable Quality Prep System helps customers accelerate their medium voltage cable preparation while embedding performance monitoring — crucial in minimizing installation time and helping customers ensure reliable energy pathways.
3M Sensored Cable Accessories provide real-time data on electrical grid performance, helping data centers detect faults, optimize energy distribution, and reduce their downtime. By integrating sensors into cable accessories, these smart grid solutions can help hyperscalers enhance their predictive maintenance, minimize outages, and improve overall grid resilience.
Optical fiber interconnects can dramatically outpace electrical copper in both power efficiency and performance. Research by Frontiers in Physics shows that optical interconnects can deliver 10 times greater energy efficiency, with improved bandwidth, lower latency, and scalability to 1.6 terabits per second and beyond. They help customers eliminate bottlenecks and enable faster real-time data exchange critical for AI compute environments. As the industry evolves, 3M will address growing AI needs beyond traditional networking with advanced solutions like active copper, dense compute fabrics for GPUs, and co-packaged optics (CPOs), positioning 3M at the forefront of materials science-based innovations.
The 3M Expanded Beam Optical Interconnect, with significantly reduced dust sensitivity compared to physical contact optical connectors, helps data centers accelerate their hyperscale deployment by reducing cleaning and inspection requirements, which accelerates time to build.
As hyperscalers target zero waste, carbon neutrality, becoming water positive, and operating on 100% renewable energy, 3M is exploring materials to help them support their sustainability goals with products such as biobased floor marking tapes, longer life and recyclable air filters and solutions to help improve water usage efficiencies. The 3M Air Containment Barrier Film provides low-carbon alternative to traditional hot-aisle containment. Effective air management is a cornerstone of efficient data center design, as strategic approaches can help significantly reduce energy overhead associated with cooling, lighting, and airflow. Furthermore, 3M vibration and noise damping solutions are helping hyberscalers mitigate nuisance noise and vibration from servers and racks.
Smart grid integration and demand flexibility
Smart grid integration is helping transform how data centers manage their electricity. Advanced, low-voltage switchboards with real-time sensing and load balancing significantly help improve resilience, reduce waste, and accommodate renewable power sources. Notably, demand response flexibility enables data centers to scale back during peak grid usage, a strategy that some advanced hyperscalers are already implementing in collaboration with local utilities.
Flexible data centers not only help utilities avoid costly grid expansions but also accelerate deployment timelines — a recent report from Energy Innovation shows that demand flexibility can reduce the need for new gas generation or grid upgrades.
Why 3M matters for AI-ready U.S. infrastructure
- Efficiency from installation to operation: By cutting cable prep time and improving reliability, 3M systems help data centers/hyberscalers reduce their labor and downtime risks.
- Next-Gen connectivity: Copper and optical interconnects help data centers/hyperscalers enable high bandwidth with low power and thermal footprint — key for dense AI workloads.
- Lower carbon solutions: 3M is committed to helping customers support their sustainability goals with innovations like biobased floor marking tapes, longer life and recyclable air filters, and solutions to help improve water usage efficiencies. Air barrier film contributes to hot aisle containment strategies that help data centers/hyperscalers meet their thermal requirements and support energy goals.
- Grid-aware infrastructure: Sensor-enabled cabling integrates seamlessly with smart grid and demand-response regimes, aligning with the U.S. aspiration to lead AI infrastructure efficiently.
As the U.S. competes on the global AI stage, data centers must be fast-built, energy-efficient, reliable, and smart-grid ready. 3M integrated solutions — from cable preparation and monitoring tools to optical interconnects and airflow management — help empower operators to meet their demands while also advancing their sustainability. Our innovations help unlock AI’s full potential with less energy, more speed, and greater resilience.
About 3M
3M (NYSE:MMM) is focused on transforming industries around the world by applying science and creating innovative, customer-focused solutions. Our multi-disciplinary team is working to solve tough customer problems by leveraging diverse technology platforms, differentiated capabilities, global footprint, and operational excellence. For more information, visit www.3m.com. Discover how 3M is shaping the future at news.3m.com/news-center.



Unlisted Expands Real Estate Network with the Courtney Stach Group in Partnership with Compass
Unlisted, a groundbreaking digital real estate community focused on the 98% of homes that aren’t for sale—yet, today announced that the Courtney Stach Group has joined the platform as a Local Expert for buyers and homeowners in Clarendon Hills, representing the 60514 ZIP code.
Unlisted’s technology is designed to open up possibilities for everyone in the real estate ecosystem: buyers, homeowners, and agents alike. Buyers gain new ways to discover homes they love beyond what’s listed on the MLS. Homeowners gain tools to better understand interest in their property and connect with potential buyers. Real estate professionals gain a program that elevates their expertise and brings more possibilities into view for their clients.
Unlisted uses publicly available data to create a digital property profile for every home in the country. These records can be quickly searched and organized into curated lists of homes that meet a buyer’s criteria. Though not for sale, buyers can join a Waitlist for their favorite homes, creating new possibilities for buyers and agents to connect and giving homeowners options they may not have considered.
Each vetted real estate professional that partners with Unlisted receives an agent profile linked to every home in their ZIP code. Only one agent is selected per ZIP, highlighting their expertise and credibility in that market. As Unlisted continues to expand this network nationwide, the goal remains the same: to provide buyers, homeowners, and agents with a platform built on connection, expertise, and meaningful insights.
The Courtney Stach Group delivers a dynamic, client-focused approach to real estate, led by Courtney Stach, who is known for her energy, intellect, and commitment to exceeding client expectations. Courtney and her team continue to evolve their hand-crafted group of preferred partners that give their clients access to an unrivaled offering of financial partners, service trades, relocation services, health providers, and more.
“Our goal is to be a true resource for clients at every stage of their journey,” said Stach. “Unlisted allows us to extend that commitment by connecting homeowners and buyers with opportunities and insights they won’t find anywhere else.”
“The Courtney Stach Group brings unmatched energy and expertise to their clients, and we’re thrilled to welcome them to Unlisted. Their client-first approach and curated partner network align perfectly with our mission to deliver meaningful connections and new opportunities.” said Katie Hill, founder and CEO of Unlisted.
To learn more about Unlisted, visit UnlistedHomes.com. For Unlisted for Agents, visit UnlistedHomes.com/Agents.
To learn more about The Courtney Stach Group, visit their Unlisted Profile or Compass.
About Unlisted
Unlisted focuses on the 98% of homes that aren’t for sale—yet. The company empowers home buyers to join the Waitlist for homes they love that aren’t for sale—in other words, unlisted. At the same time, homeowners collect a Waitlist of interested buyers for whenever the time comes to sell, giving everyone a head start. Unlisted also allows homeowners to control how their home is presented online; they can create a stunning up-to-date property profile that shows off the home’s best features and attracts more interest. With more time and more connection, Unlisted is a more human way to explore real estate that drives better outcomes for all. For more information, visit UnlistedHomes.com.
Media Contact
Sophia Jacomet
sophia@unlistedinc.com



Redstone Consultants OÜ Launches Cashivo.com, a Digital Gift Card Platform
Redstone Consultants OÜ has launched Cashivo, a secure digital platform designed to modernize the way electronic gift cards are accessed and managed. The service has been created to meet the increasing demand for simple and reliable digital solutions in Europe. Unlike traditional schemes with complex procedures, Cashivo is built to deliver speed and simplicity. Transactions are processed in real time, and balances are updated instantly in users' accounts.
Platform Features
Cashivo combines convenience with accessibility through a user experience designed for digital-first markets. Key features include:
- Cross-Brand Use: Gift cards can be applied across a range of international retailers.
- Fast Onboarding: Accounts can be created and activated within minutes.
- Streamlined Interface: Navigation and management are simplified through intuitive design.
Compliance and Security
Operating under EU regulatory standards, Cashivo prioritizes transparency and user protection. The platform integrates encryption protocols to secure every transaction and safeguard personal data.
"Cashivo eliminates much of the complexity associated with traditional programs," said Silver Karutoom, director of Cashivo. "Our focus has been on creating a solution that is both secure and easy to use."
Company Vision and Market Outlook
Headquartered in Tallinn, Estonia, Redstone Consultants OÜ draws on the country's advanced digital infrastructure and fintech ecosystem. With the launch of Cashivo, the company aims to strengthen its role in Europe's growing digital services sector. The roadmap includes expanding brand partnerships, introducing new categories of digital rewards, and incorporating user feedback into platform updates.
Cashivo is now available to users across Europe.
About Redstone Consultants OÜ
Redstone Consultants OÜ is a technology company registered in Estonia and headquartered in Tallinn. The company develops secure, user-friendly fintech platforms, with a focus on compliance and accessibility. Cashivo is its latest product, aimed at delivering a streamlined approach to digital gift card management. Learn more at www.cashivo.com.
Media Contact
Neil Allan Pool
support@cashivo.com



MadMuscles Launches Free Adaptive Training Program for Wounded Veterans and People with Disabilities
The MadMuscles app has recently announced the launch of a new free program of adaptive training for individuals with amputations and limited mobility, veterans who sustained injuries as a result of military actions, as well as people who recover from surgeries or other injuries.
The program was created in cooperation with Ihry Neskorenykh in Ukraine (the Invictus Games community in Ukraine). Both professional coaches and team members took part in the program development. The MadMuscles adaptive program focuses on individuals who suffer from damaged limb functionality, spinal cord injuries, and war veterans who have finished the initial rehabilitation stage and are striving to get back to an active lifestyle.
What Makes the MadMuscles Workout Program the Best Choice for People with Disabilities and Injured Veterans: Top Facts
Starting the new training routine can be overwhelming, but MadMuscles has done its best to make the adaptive program simple, safe, and motivating:
- All you need to start free adaptive training is to download the MadMuscles mobile app and complete the short quiz to determine the workout routine that will work best for you.
- The program was developed by professional coaches who take part in the veterans’ rehabilitation and veterans from the community of Ihry Neskolenykh in Ukraine (the Invictus Games community in Ukraine) themselves, who have completed the first stage of rehabilitation
- The training program is divided into 6 categories based on different types of injuries and conditions.
- Exercises don’t require any special equipment and are designed to train whenever, wherever, and however you want and can.
- Users can also explore motivational content from former participants of the Invictus Games and adaptive sports influencers who will share their stories and experiences of rehabilitation.
- The MadMuscles app is available for both iOS and Android operating systems and doesn’t involve any complicated registration procedures.
Introduction to Adaptive Training: Workouts Based on the Capabilities of Your Body
The key peculiarity of adaptive programs is that they take into account the needs of people with amputations, spinal cord injuries, or other issues. MadMuscles believes that creating a program in accordance with the real capabilities of users makes it much more efficient when it comes to recovering balance, power, and stamina. With this, it gets easier to regain confidence and get a boost of motivation to keep an active lifestyle.
In addition to being adaptive in terms of physical abilities, the MadMuscles workout program also takes into account that people after rehabilitation can have limited access to different resources. Thus, MadMuscles training can be performed at home without any special gym equipment. Once the medical rehabilitation is complete, the user can safely get down to physical training.
Who Can Use the Program
What makes MadMuscles stand out from the crowd of other training apps is that it goes beyond basic fitness routines. The adaptive program is very specialized, and the professional coaches worked on it to make it suitable for six categories. So, the MadMuscles program is made for everybody from the following categories:
- Combined impairment: One or both lower limbs + one upper limb.
- Both arms: Double amputation or equivalent impairment.
- Both legs: Spinal cord injury, double amputation, or equivalent impairment.
- One arm: Amputation or functional impairment.
- One leg: Amputation or functional impairment.
- Preserved functionality: All limbs are partially or fully functional, and physical activity is possible.
All six categories will have access to safe and effective fitness routines tailored to their needs, along with inspirational stories of the veterans from the community of Ihry Neskorenykh in Ukraine (the Invictus Games community in Ukraine) and adaptive sports influencers.
Partnership with the Invictus Games community in Ukraine: How Coaches and Participants Helped to Develop the Program
For MadMuscles, authentic insights from those who know first-hand all the challenges of returning to an active lifestyle after suffering injuries became vital in creating the adaptive program. So, the team drew on the experience of the Invictus Games community in Ukraine, an initiative that aims to help war veterans come back to an active lifestyle through competition and adaptive sports. Those people know the pitfalls of the rehabilitation path better than anyone else. Only those who have a similar combat and athletic experience can estimate what is most effective for veterans and what will correspond with their actual needs.
By involving assistance from real war veterans who have suffered severe injuries and come back to an active lifestyle through sports, the MadMuscles aim to embrace expertise and trust. It is also vital to showcase these real examples to inspire users of the app and make them understand that nothing is impossible if you have the true inner strength!
More than Just Training: The Power of Motivation
Everyone who has been trying to go in for sports knows that you need not only physical ability to do exercises but also enough motivation to keep going. People who have suffered amputations or mobility limitations are often in a rather difficult mental state. They have to deal with immense psychoemotional pressure as they need to overcome insecurity, fear, and other psychological barriers to get a start.
All those challenges of a new reality influence the workout routine far more than most people realize. However, other fitness apps hardly pay attention to them. MadMuscles is going to change that perspective once and forever.
MadMuscles isn’t just a set of effective adaptive exercises but also a digital library of motivational content. The developers believe that such an approach inspired the program with a deeper meaning than just exercising, as it establishes real emotional ties between the app and users.
The most important thing about motivation within the app is that it introduces real stories of veterans who have already gone the same way and overcome the same difficulties. Their video stories will be a perfect showcase that even in the most difficult situations, a person with a strong will can achieve fitness goals. Injury isn’t a limitation for your strength, both inner and physical. Each video story delivers a simple yet real-life-based message: you can train, develop your power, and gain muscle mass — MadMuscles will guide you through this process effectively.
How to Start Training with the MadMuscles App
The top objective of MadMuscles was to make the program easy to use. That’s why the developers made its start as simple as possible. Complete these five simple steps on your way to an active lifestyle through adaptive training:
- Download the MadMuscles mobile app from the Google Play or App Store.
- Complete the adaptive quiz by specifying your physical conditions and the peculiarities of your injury to get a program that is tailored to your needs.
- Read the instructions on safety measures.
- Start your training whenever you are ready and keep track of your progress in real time.
- Watch inspirational video stories of the members of the community of Ihry Neskorenykh in Ukraine (the Invictus Games community in Ukraine) and adaptive sports influencers.
MadMuscles Broadens the Inclusive Fitness Borders and Eliminates Barriers
Social inclusivity and digital adaptability are the key focuses of the MadMuscles adaptive program for individuals with amputations and war veterans. The developers of the program believe that they achieve this by completing the following roadmap:
- The adaptive program will be available in different languages, including English, Italian, Spanish, French, German, Brazilian Portuguese, and Ukrainian, to ensure that the objective of providing efficient adaptive training will be fulfilled worldwide.
- The program is free and is developed with the help of professional coaches and those who have faced similar conditions resulting from their participation in combat.
- The MadMuscles app not only enables efficient training but also inspires people to become more confident in themselves through motivating stories of the veterans from the community of Ihry Neskorenykh in Ukraine (the Invictus Games community in Ukraine) and adaptive sports influencers.
About MadMuscles
MadMuscles is a mobile fitness app offering diverse training programs, personalized coaching, and healthy meal plans. The app also provides a free adaptive training program designed to support people with disabilities and limited mobility. Created by AmoApps Limited, MadMuscles reflects the company’s commitment to developing effective, innovative products powered by technologies such as AI and habit-forming tools that help users achieve lasting progress. The program is available worldwide on both Android and iOS. Learn more at madmuscles.com/adaptive-workout-program.
Media Contact
John Peterson
Blog writer and professional fitness coach, AmoApps Limited
feedback@madmuscles.com
.jpeg)
.jpeg)

Experience the Future of Manufacturing at KMM Group’s MFG Day Event
KMM Group, Ltd. will open its doors to the community for Manufacturing (MFG) Day 2025 on Friday, Oct. 3, offering a firsthand look at the world of advanced manufacturing. Students, educators and community members are invited to take part in guided tours of KMM’s 100,000-square-foot state-of-the-art facility and explore how today’s innovations are shaping the future of industries such as medtech and aerospace.
During each guided tour, participants will see high-tech equipment that produces lifesaving components with ultra precision. Along the way, machinists and engineers will demonstrate processes such as milling, turning and grinding, providing a behind-the-scenes look at how ideas become real-world solutions. The event is designed to bring STEM to life, showing students and community members how rewarding and dynamic manufacturing careers can be in today’s economy.
“By opening our doors on MFG Day, we aim to teach, inspire, enlighten and amaze those who will take the time to participate,” said John Shegda, CEO of KMM Group. “Many will have no idea of the impact that a company like ours has within our community and, upon people around the world, when they walk through our doors. They will leave with a much stronger understanding of the importance of manufacturing and of the incredible things that we make possible through the science of engineering and combining it with passion and creativity."
The event will take place Friday, Oct. 3, 2025, at KMM Group’s facility at 2200 Byberry Road in Hatboro, Pennsylvania. Tours are scheduled from 8 a.m. to noon, each lasting about 45 minutes.
Educators may register their classes, and community workforce leaders may register their groups by visiting mfgday.com or KMM Group’s website. Space is limited, so early registration is encouraged. The event is free to attend, and members of the media are invited to capture b-roll and interview participants and company leaders.
About KMM Group
KMM Group, Ltd., is a collaborative fusion of three world-class companies: KV Inc., M&S Centerless Grinding, Inc. and Meron Medical. It manufactures complex components for medical, aerospace, space exploration, high-tech and defense industries, drawing on its collective 100-year history of leading-edge ultra-precision machining and centerless grinding experience. For more information, visit kmmgrp.com.
About MFG Day
MFG Day is a national initiative of The Manufacturing Institute designed to highlight modern manufacturing, inspire the next generation of makers, and address workforce needs across the country. For more information, visit mfgday.com.
Media Contact
Amy Rodgers
amyr@kmmgrp.com



Unlisted Expands Network of Real Estate Professionals with Brittany Baity of Compass
Unlisted, a groundbreaking digital real estate community focused on the 98% of homes that aren’t for sale—yet, today announced that Brittany Baity of Compass has joined the platform as a local expert for buyers and homeowners in Dallas, representing the 06830, 06831, 06807, 06878, and 06870 ZIP codes.
Unlisted’s technology is designed to open up possibilities for everyone in the real estate ecosystem: buyers, homeowners, and agents alike. Buyers gain new ways to discover homes they love beyond what’s listed on the MLS. Homeowners gain tools to better understand interest in their property and connect with potential buyers. Real estate professionals gain a program that elevates their expertise and brings more possibilities into view for their clients.
Unlisted uses publicly available data to create a digital property profile for every home in the country. These records can be quickly searched and organized into curated lists of homes that meet a buyer’s criteria. Though not for sale, buyers can join a Waitlist for their favorite homes, creating new possibilities for buyers and agents to connect and giving homeowners options they may not have considered.
Each vetted real estate professional that partners with Unlisted receives an agent profile linked to every home in their ZIP code. Only one agent is selected per ZIP, highlighting their expertise and credibility in that market. As Unlisted continues to expand this network nationwide, the goal remains the same: to provide buyers, homeowners, and agents with a platform built on connection, expertise, and meaningful insights.
Brittany Baity offers clients both business savvy and local insight, drawing on her background in high-level negotiations and nearly a decade of living in Greenwich. Detail-oriented and proactive, she helps families navigate the transition from city to suburban life with ease, providing first-hand knowledge of schools, neighborhoods, and community life.
“I’m excited to bring this innovative approach to the Greenwich area and help people discover what’s possible beyond the traditional market,” said Baity.
“We’re proud to welcome Brittany Baity as our Local Expert in these Greenwich ZIP codes,” said Katie Hill, founder and CEO of Unlisted. “Her local knowledge and thoughtful approach to real estate make her an incredible addition, and we know she’ll create opportunities that reflect the strength and character of this community.”
To learn more about Unlisted, visit UnlistedHomes.com. For Unlisted for Agents, visit UnlistedHomes.com/Agents.
To learn more about Brittany Baity, visit her Unlisted Profile or her website.
About Unlisted
Unlisted focuses on the 98% of homes that aren’t for sale—yet. The company empowers home buyers to join the Waitlist for homes they love that aren’t for sale—in other words, unlisted. At the same time, homeowners collect a Waitlist of interested buyers for whenever the time comes to sell, giving everyone a head start. Unlisted also allows homeowners to control how their home is presented online; they can create a stunning up-to-date property profile that shows off the home’s best features and attracts more interest. With more time and more connection, Unlisted is a more human way to explore real estate that drives better outcomes for all. For more information, visit UnlistedHomes.com.
Media Contact
Sophia Jacomet
sophia@unlistedinc.com



Abu Dhabi DMT and EchoTwin AI Partner to Launch Smart Sweep AI, Pioneering the Future of Cognitive Cities
Abu Dhabi Department of Municipalities and Transport (DMT) has partnered with EchoTwin AI, a leader in AI-powered urban intelligence, to launch Smart Sweep AI—a transformative initiative that turns everyday municipal street-sweeping fleets into real-time urban intelligence platforms. This collaboration reflects a shared vision to position Abu Dhabi as a global model for the next generation of cognitive cities—urban environments that can sense, interpret, and act intelligently to improve quality of life, resilience, and sustainability.
Through Smart Sweep AI, DMT is deploying EchoTwin AI’s CityVision One edge devices and CityWide platform across its sweeping vehicles, transforming them into intelligent sensing units that continuously monitor road conditions, cleanliness, and compliance. Powered by EchoTwin AI’s proprietary Vision-Language Model Framework (VLMF), the system can detect issues ranging from litter accumulation and overflowing bins to broken infrastructure and safety hazards—triggering automated reporting and workflows that accelerate resolution.
“Smart Sweep AI is a first-of-its-kind project in the region, positioning Abu Dhabi at the forefront of data-driven municipal services,” said Chris Carson, founder and CEO of EchoTwin AI. “We are redefining how cities operate—embedding See,Think, Act intelligence into everyday fleets so that cities themselves become aware and responsive. This is how cognitive cities are born: cleaner streets, safer communities, and sustainable environments achieved through urban intelligence at scale.”
Since its initial deployment, Smart Sweep AI has delivered tangible outcomes. Across a limited fleet, the system recorded more than 52,000 assets and identified nearly 20,000 compliance issues in just 60 days—evidence of the scale and accuracy made possible by augmenting everyday operations with AI-driven sensing.
This initiative directly supports Abu Dhabi’s smart city and sustainability strategies, building a proactive compliance framework that enhances transparency, accelerates issue resolution, and sets new benchmarks for urban livability.
“DMT’s mission is to achieve the highest global standards of livability and sustainability,” said a DMT spokesperson. “With Smart Sweep AI, we are moving from reactive inspections toward proactive, AI-driven compliance across the emirate. This step accelerates Abu Dhabi’s transformation into a smarter, cleaner, and more resilient global city.”
EchoTwin AI will showcase the Smart Sweep AI at GITEX Global 2025 in Dubai, Sept. 13–17, where visitors can experience the platform’s See, Think, Act intelligence—demonstrating how AI-powered fleets can reshape municipal services and pave the way for cognitive cities.
About EchoTwin AI
EchoTwin AI is a leader in infrastructure intelligence, redefining how cities are managed. Powered by a proprietary visual intelligence engine with full spatial reasoning, EchoTwin transforms municipal fleets into mobile urban sensors—creating living digital twins that provide real-time insights into infrastructure, compliance, and safety. By enabling municipalities to proactively monitor, predict, and resolve issues, EchoTwin helps build resilient, self-healing, and sustainable urban ecosystems. More than “smart cities,” EchoTwin is advancing the era of cognitive cities—urban environments with the awareness to see, think, and act on challenges in real time. Learn more at www.echotwin.ai.
Media Contact
Chris Carson
CEO, EchoTwin AI
chris.carson@echotwin.ai



Press Earth Expands Platform to Redefine Global Media Visibility
Press Earth today announced the expansion of its global public relations platform, designed to transform how brands secure international media visibility.
In an increasingly competitive marketplace, the platform helps companies turn brand stories into editorial-style features that appear in respected global publications — simplifying access, amplifying impact and strengthening corporate reputation.
Every day, thousands of press releases are distributed, yet most fail to achieve meaningful impact. Press Earth addresses this gap by reshaping brand announcements into editorial narratives tailored for international audiences and placing them within trusted global media environments. This approach allows companies not only to be visible but also to earn credibility and remain memorable on a global scale.
Visibility in globally recognized outlets extends far beyond awareness. It reinforces corporate trust, attracts investors and strategic partners, and positions brands as leaders within their industries. By placing storytelling at the center of communications, Press Earth empowers businesses to express themselves with greater authority and clarity on the world stage.
The company’s mission is to democratize global media visibility. Through a streamlined model that combines worldwide reach with editorial storytelling, Press Earth enables companies of all sizes — from startups to Fortune 500 firms — to build trust, expand internationally and control their narrative across borders.
About Press Earth
Headquartered in the U.S., Press Earth brings together the credibility of editorial journalism with the scale of global distribution. The platform helps organizations strengthen reputation, attract partnerships, and grow their influence worldwide. To learn more, visit www.pressearth.com.
Media Contact
Press Earth
clairedawson@pressearth.com



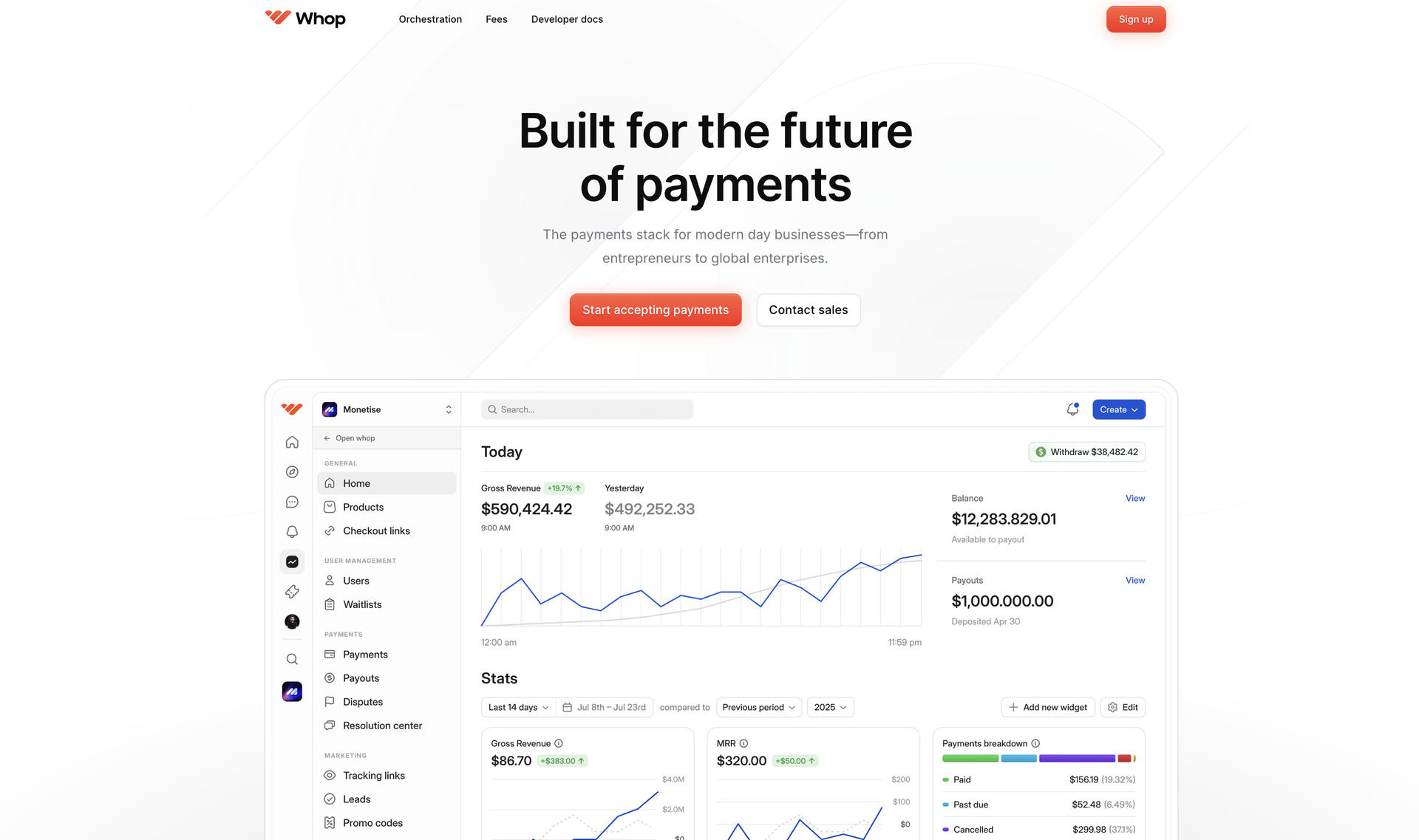
Whop Revolutionizes Online Payments with Smart Routing Technology
Whop, a social commerce platform, launched a comprehensive payment infrastructure, Whop Payments, that connects to multiple processors simultaneously, automatically rerouting declined transactions to alternative providers while offering sellers instant payouts via cryptocurrency and traditional banking channels across 241 territories worldwide.
The new payment infrastructure represents the company's transition from relying on external payment services to operating its own complete system. The platform orchestrates transactions across various payment service providers, redirecting payments in real-time when initial processing attempts fail, ensuring checkout completion without customer interruption.
Sellers accessing the new infrastructure can accept payments through multiple processors while maintaining single integration and unified reporting. The system's backup routing activates instantly when transactions encounter declines, routing payments through alternative processors to maintain sale completion rates.
Payout flexibility stands as a key component of the infrastructure. Sellers receive funds through their preferred channels — traditional bank transfers, Bitcoin, stablecoins, or digital wallets like Venmo. Local payment networks across 241 territories enable instant payouts, while cryptocurrency options provide alternatives for sellers in regions with banking limitations.
The platform incorporates native buy-now-pay-later functionality, integrated identity verification for rapid seller onboarding, and transparent flat-rate pricing across all transaction types. These features eliminate the need for sellers to establish relationships with multiple service providers or navigate complex fee structures.
Implementation accommodates various business models through embedded checkout components, standalone payment links, or full platform integration. Sellers transitioning to the new infrastructure require no additional technical setup if already using Whop's services.
Early performance data shows substantial improvements in transaction approval rates, with sellers reporting fewer abandoned checkouts and increased revenue from previously lost sales. International sellers particularly benefit from the multi-processor approach, which bypasses regional restrictions and processor-specific limitations.
The infrastructure development reflects growing demand for payment flexibility among digital entrepreneurs. As online businesses expand globally, traditional single-processor setups increasingly fail to meet diverse customer payment preferences and regional requirements.
Whop currently serves hundreds of thousands of sellers, primarily in the creator economy sector, having processed $1.5 billion in total transaction volume. The company's seller base includes educators, community builders, and digital product sellers operating subscription-based and one-time purchase models.
The rollout positions Whop to capture additional market share in the competitive digital payments space, where differentiation increasingly depends on authorization rates and payout flexibility rather than basic processing capabilities.
Steven Schwartz, co-founder at Whop, explains it simply:
“We’ve built orchestration to route every single payment to a number of different service providers in order to max out authorization rate.”
“Sellers in every country can now get paid out via local banking rails, Bitcoin and stablecoin. With this foundation in place, we’ll be laser‑focused on structuring how people actually make money on the internet to ensure sustainable income for everybody.”
Hunter Dickinson, head of partnerships at Whop, adds:
“When we started, Whop Payments was essentially a Stripe Connect wrapper. Today, we’ve built our own infrastructure — from KYC to pay-ins and payouts. If a transaction fails on one processor, we automatically reroute it behind the scenes so the customer never notices. That means higher payment acceptance rates, smoother checkouts, and fewer lost sales for our customers. On top of that, we can pay creators out however they want through our payout partner— whether it’s in Bitcoin, Venmo, or more.”
“Through thousands of conversations, we’ve learned businesses really only care about two things: getting paid and paying out. That means rates, payment acceptance, failed transactions, and fees. Our mission is to be the best in the world at solving those problems.”
“At Whop, we build by obsessing over our customers. Payments aren’t just a feature — they’re the foundation of every business online. So instead of guessing, we ask, listen, and iterate until we deliver the payment solutions that actually move our customers’ businesses forward.”
About Whop
Whop is a social commerce platform for online entrepreneurs. Instead of stitching together multiple tools, creators can run everything from courses and communities to payments, marketing, and support in one place: their own online hub, or “whop.” Trusted by hundreds of thousands worldwide, Whop has already helped generate over $1.5 billion in revenue — making it the go-to platform for turning passions into income. For more information, visit whop.com.
Media Contact
Karina Egle
Whop
karina@whop.com


Dr. Carl June and Dr. Michel Sadelain to Receive Broermann Medical Innovation Award 2025
The jury of the Broermann Medical Innovation Award is honoring Dr. Carl June of the University of Pennsylvania and Dr. Michel Sadelain of Columbia University for their groundbreaking research in CAR-T cell therapy. The award is being presented for the first time this year. With prize money of 1 million euros, it is among the most highly endowed awards for medical research worldwide.
A Revolution in Cancer Therapy
June and Sadelain are recognized for their pioneering roles in the genetic modification of T cells, enabling them to identify, attack and destroy cancer cells. They use synthetic receptors, called chimeric antigen receptors (CARs), which allow T cells to target and kill cancer cells. CAR-T cell therapies have achieved remarkable success in treating cancers such as leukemia, lymphoma and myeloma. For other cancers, the approach remains in the experimental phase.
June, the Richard W. Vague Professor in Immunotherapy at the University of Pennsylvania’s Perelman School of Medicine, said:"I am deeply honored to receive the inaugural Broermann Medical Innovation Award together with my esteemed colleague Michel Sadelain. This award not only recognizes our decades of research but also the transformative potential of CAR-T cell therapy for cancer patients worldwide."
Sadelain, director of the Columbia Initiative in Cell Engineering and Therapy, said: "Receiving the Broermann Medical Innovation Award is an extraordinary honor and recognition of the revolutionary power of CAR-T cell therapy. Together with Carl June, we have worked to turn the immune system into a precise weapon against cancer. Our work reflects what the Broermann Award stands for: medical breakthroughs that fundamentally change patients’ lives. It fills me with profound gratitude to know that our research helps usher in a new era of cancer treatment and creates hope for patients once considered incurable."
CAR-T cell therapy has achieved remarkable success rates in treating cancers such as leukemia, lymphoma and myeloma. Through their visionary research, Sadelain and June have ushered in a completely new era in cancer treatment.
Selection Process by Renowned Jury
Laureates of the Broermann Medical Innovation Award are selected by a scientific jury of nine senior members of leading institutions: Prof. Dr. Werner Seeger (chair of the award), Prof. Dr. Karsten Krüger (Justus Liebig University Giessen), Prof. Dr. Isabelle Bekeredjian-Ding (University of Marburg), Prof. Dr. Britta Siegmund (German Research Foundation), Prof. Dr. Stefan Offermanns (Max Planck Institute for Heart and Lung Research), Prof. Dr. Otmar Wiestler (Helmholtz Association), Prof. Dr. Dr. Gerd Geißlinger (Fraunhofer Institute for Clinical Pharmacology), Prof. Dr. Martina Brockmeier (Leibniz Association) and Dr. Jan Liersch (Broermann Holding GmbH). Supported by international reviewers, the committee evaluates nominations in a multi-stage process based on strict selection criteria to ensure scientific quality and independence.
About the Laureates
Carl June was born in Denver in 1953. He received his medical degree from Baylor College of Medicine in Houston and completed postdoctoral training at the Fred Hutchinson Cancer Research Center in Seattle. Following a research career with the U.S. Navy, he founded the Center for Cellular Immunotherapies at the University of Pennsylvania in 1999. In 2012, he received the Richard W. Vague Professorship.
Michel Sadelain was born in Paris in 1960. He received his medical degree from the University of Paris in 1984 and his doctorate from the University of Alberta in 1989, followed by postdoctoral research at the Whitehead Institute for Biomedical Research at the Massachusetts Institute of Technology. In 1994, he joined Memorial Sloan Kettering Cancer Center in New York. In 2024, he became director of the Columbia Initiative in Cell Engineering and Therapy.
Both scientists have received numerous honors for their contributions to cancer therapy.
About the Broermann Medical Innovation Award
The Broermann Medical Innovation Award was established in 2024 by Dr. Bernard große Broermann, founder of the Asklepios Kliniken Group. Throughout his career, große Broermann pursued the vision of building an innovative health care company that delivers lasting value to patients. Under his leadership, Asklepios set new standards in medical innovation, investing in advanced technologies and digital solutions to improve care.
The award continues his vision by recognizing pioneering medical achievements. With a prize of 1 million euros, it is presented at the Hessian State Chancellery in Wiesbaden by Hessian Prime Minister Boris Rhein.
For more information, visit www.broermann-award.org.
Media Contact
Miriam Malko
contact@broermann-award.org
+49 641 98542310
Asklepios Kliniken Konzernbereich Unternehmenskommunikation & Marketing
presse@asklepios.com
+49 40 1818826636
24-hour press office hotline
presse@asklepios.com
+49 40 1818828888



Online Betting Growth in the UK: Can the Industry Continue to Grow?
The UK’s online casino sector entered 2025 at a crossroads: still expanding, but doing so under tighter regulatory oversight and rising public scrutiny.
What began as a pandemic-era boom in mobile play has evolved into a more complex market shaped by product changes, sharper consumer-protection rules, shifting player habits and diverging industry forecasts.
Market snapshot: steady growth, mixed forecasts
Analysts and market researchers broadly agree the UK’s online gambling market remains substantial and is expected to grow further through the decade, but projections vary on speed and scale.
Some industry projections show a relatively high compound annual growth rate for the online market into the late 2020s, pointing to continued expansion driven by mobile adoption, online sports betting and game innovation.
At the same time, official UK data present a more nuanced picture: participation rates for online gambling are sizeable (with many consumers taking part in lotteries, sports betting and casino games online), and the Gambling Commission’s operator-reported figures continue to highlight the financial importance of remote (online) verticals to licensed operators.
Regulatory shifts are reshaping product design and limits
2024–2025 saw a steelier regulatory hand from Westminster and the Gambling Commission, and those interventions are now feeding directly into the online casino product roadmap.
New remote game design rules and player protection measures introduced in 2024 and refined into 2025 have forced operators to redesign commonly used features — for example, limits on autoplay, restrictions on rapid-play mechanics, minimum spin speeds and requirements to display net spend and time information for casino play.
Those changes have two immediate consequences.
First, certain forms of high-frequency play that previously drove session length and short-term revenue are being phased out or curbed, trimming what had been a straightforward growth lever for operators.
Second, compliance costs have risen as operators update game code, user interfaces and monitoring systems to meet new financial vulnerability-check thresholds and safer-play messaging requirements.
Player behaviour: mobile, money management and demographics
Mobile remains the dominant channel for casino play in the UK, and game studios continue to optimise for smaller screens and fast, intuitive UX.
But beyond access, behaviour is shifting: operators and regulators alike report more demand for clearer spend information, voluntary limits, and tools that help players monitor time and losses.
Demographically, while older cohorts still engage with lotteries and betting, the core online casino players tend to be younger adults who favour slots, instant-win and live-dealer formats when seeking entertainment rather than pure wagering.
The Gambling Commission’s participation surveys through 2023–24 highlighted how a large share of online activity is lottery-related, but when lottery-only players are excluded the profile tilts younger and more digitally native.
Operator strategies: product, consolidation and partnerships
Faced with new product rules and higher compliance costs, many operators pivoted in 2025 toward diversification and premiumisation.
That means more emphasis on regulated live-casino offerings, skill-based or hybrid games that can be framed as entertainment, and partnerships with regulated game studios that will meet the UK’s design requirements out of the box.
Consolidation has also accelerated: where margins permit, larger groups are acquiring niche brands and technology vendors to control supply chains and reduce unit compliance costs.
At the same time, some smaller or offshore-adjacent operators are finding the UK market’s compliance burden and enforcement climate less hospitable, which clears room for licensed, compliance-focused operators to capture market share.
Advertising, taxes and public scrutiny — headwinds for growth
Online casino growth isn’t operating in a vacuum.
Political and media attention on problem gambling, the taxation of online play and advertising standards are real headwinds.
Public debates in 2024–25 included calls for higher taxes on gambling revenue and closer controls on promotional practices, and prominent voices suggested fiscal or regulatory responses that could alter operator economics if implemented.
Tighter advertising controls and the reputational risk of high-profile enforcement actions also mean marketing strategies are evolving: fewer flashy acquisition promotions and more on-site retention and product-bundling tactics that lean on lifetime value rather than short-lived sign-up spikes.
Outlook: measured expansion, more durable market
Overall, the UK online casino market in 2025 looks set for measured — not runaway — growth.
Mobile adoption, improved game experiences and a maturing regulatory framework mean the sector should remain commercially important and resilient.
But growth will be slower and structurally different than the disruption-driven expansion of earlier years.
Operators who invest in compliant product design, financial safeguards, transparent player tools and diversified revenue models are best positioned to win customers and regulators’ trust alike.
Conversely, those who treat compliance as a cost line item rather than a strategic differentiator may struggle as enforcement tightens and consumer expectations rise.
What to watch next
In the near term, three things will determine whether growth accelerates or stalls: the shape of any new tax measures, further rules on game design and advertising, and how effectively operators can monetise safer, slower-play products without sacrificing lifetime value.
For consumers, the promise is clearer protections and more transparent play.
For industry stakeholders, the task is to translate entertainment into sustainable, regulated products that survive scrutiny — and still deliver enjoyable games.



Unlisted Expands Network of Real Estate Professionals with Jason Clark of Briggs Freeman Sotheby’s International Realty
Unlisted, a groundbreaking digital real estate community focused on the 98% of homes that aren’t for sale—yet, today announced that Jason Clark of Briggs Freeman Sotheby’s International Realty has joined the platform as a local expert for buyers and homeowners in Dallas, representing the 75206 and 75214 ZIP codes.
Unlisted’s technology is designed to open up possibilities for everyone in the real estate ecosystem: buyers, homeowners, and agents alike. Buyers gain new ways to discover homes they love beyond what’s listed on the MLS. Homeowners gain tools to better understand interest in their property and connect with potential buyers. Real estate professionals gain a program that elevates their expertise and brings more possibilities into view for their clients.
Unlisted uses publicly available data to create a digital property profile for every home in the country. These records can be quickly searched and organized into curated lists of homes that meet a buyer’s criteria. Though not for sale, buyers can join a Waitlist for their favorite homes, creating new possibilities for buyers and agents to connect and giving homeowners options they may not have considered.
Each vetted real estate professional that partners with Unlisted receives an agent profile linked to every home in their ZIP code. Only one agent is selected per ZIP, highlighting their expertise and credibility in that market. As Unlisted continues to expand this network nationwide, the goal remains the same: to provide buyers, homeowners, and agents with a platform built on connection, expertise, and meaningful insights.
For the past 18 months, Clark has expanded his services through Unlisted, working closely with the team and helping influence the company’s growth and direction. His deep involvement shows his commitment to bringing new possibilities to buyers and homeowners in East Dallas and Lakewood.
“Unlisted is opening new doors in real estate, giving buyers and homeowners insights they’ve never had before,” said Clark. “I want to be a resource for anyone in the 75206 and 75214 ZIP codes who’s curious about their options, whether they’re looking to buy, exploring selling, or simply want to understand what’s possible.”
“We’re excited to have Jason Clark continuing to grow with us in Dallas,” said Katie Hill, founder and CEO of Unlisted. “Jason has been part of our journey for more than a year, helping us expand our vision and bring real value to buyers and homeowners. His knowledge of 75206 and 75214 makes him the ideal partner for Unlisted.”
To learn more about Unlisted, visit UnlistedHomes.com. For Unlisted for Agents, visit UnlistedHomes.com/Agents.
To learn more about Jason Clark, visit his Unlisted Profile or his website.
About Unlisted
Unlisted focuses on the 98% of homes that aren’t for sale—yet. The company empowers home buyers to join the Waitlist for homes they love that aren’t for sale—in other words, unlisted. At the same time, homeowners collect a Waitlist of interested buyers for whenever the time comes to sell, giving everyone a head start. Unlisted also allows homeowners to control how their home is presented online; they can create a stunning up-to-date property profile that shows off the home’s best features and attracts more interest. With more time and more connection, Unlisted is a more human way to explore real estate that drives better outcomes for all. For more information, visit UnlistedHomes.com.
Media Contact
Sophia Jacomet
sophia@unlistedinc.com
.jpeg)
.jpeg)

RIIZE’s Wonbin Highlights TOOMUCHTAX 'Printed Waffle' Set at Incheon Airport
Seoul-based contemporary fashion brand TOOMUCHTAX announced that Wonbin, a member of the K-pop boy group RIIZE, was seen Aug. 21 at Incheon International Airport wearing the brand’s “Printed Waffle Zip-up Hoodie (Ivory)” and “Printed Waffle Pants (Ivory).”
The set, recognized for its vintage ivory tone and waffle-knit texture, quickly drew attention from fans and fashion observers. Following the appearance, TOOMUCHTAX reported a surge in online orders, with the items selling out in multiple sizes.
“We are grateful for the enthusiastic response,” a TOOMUCHTAX spokesperson said. “To meet growing demand, we have initiated an urgent re-order, and the items will be restocked Sept. 19. Customers can also purchase the Printed Waffle Zip-up Hoodie in gray, which remains available through our official store.”
TOOMUCHTAX has emerged as a rising K-fashion label worn by K-pop idols and global celebrities. Wonbin’s airport look further underscores the brand’s momentum ahead of its fall/winter 2025 collection.
About TOOMUCHTAX
TOOMUCHTAX, based in Seoul, South Korea, draws inspiration from everyday life, creating designs that combine vintage charm with sensory elegance. Each collection reflects the brand’s philosophy that “beauty is individuality,” celebrating the unique allure of women. From ready-to-wear pieces that embrace the body to streetwear with refined elegance, TOOMUCHTAX offers styles that highlight natural beauty through quality craftsmanship and a thoughtful mix of textures. For more information, visit toomuch.tax.
Media Contact
Seulbeom Park
Online Media Manager
sbsiriai@gmail.com




Turn your news into performance
EZ Newswire is the only news platform connecting the most influential organizations with the most trusted media outlets.