Newsroom
Official news releases and announcements from organizations worldwide, distributed by EZ Newswire.
Cardtonic CEO Emmanuel Sohe Shares Blueprint for Sustainable Revenue at Web Summit Vancouver 2026
Cardtonic, a leading African fintech platform serving over 1.8 million active users across Nigeria and Ghana, announced the participation of its chief executive officer, Emmanuel Sohe, in the "The New Go-To-Market Blueprint: Building Predictable Revenue in 2026" panel session at Web Summit Vancouver 2026. The global tech conference is held between May 11 and 14 at the Vancouver Convention Centre, Canada.
Sohe was among the invited operators who shared practical, experience-backed insights on building predictable and sustainable revenue. What made Cardtonic’s story stand out is that the company reached this scale without raising a single dollar of institutional seed capital.
Building Predictable Revenue in an Emerging Market
Sohe opened his contribution by putting the challenge in context. “If you are from an emerging market where I am from, where trust, infrastructural constraints, and regulatory complexity make it a bit difficult to predict your revenue, you are quick to learn that you need more than just a blueprint to scale in the market,” he told the audience.
For Cardtonic, operating in a volatile macroeconomic environment forced a level of structural discipline that most well-funded startups never develop. Growth was tied to actual customer demand rather than vanity metrics like total downloads, page views, or registered user counts.
That discipline is visible in how the business scaled: from a manual gift card trading operation to a multi-product super-app offering virtual dollar cards, NFC contactless cards, eSIMs, bill payment services, and a gadget store, all funded by cash-flowing utility rather than outside capital.
Three Things Cardtonic Says Every Product Needs
Sohe outlined three practical requirements he believes any product must meet to generate predictable revenue, particularly in the African market.
“One, your product needs to be innovative. What are you bringing to the market? Can your users depend on it? You can come up with all of the frameworks and models you need, but without an innovative product, there’s a high chance that you’ll still fail. Two, pricing is a big deal back home in Africa. People are very careful about their one cent. So your pricing also has to be competitive. Third, your tech needs to be agile. Your engineering velocity is almost proportionally related to your revenue velocity,” Sohe said.
The Role of Product in Revenue Strategy
The panel also explored whether siloed departments hurt or help a startup’s ability to generate consistent revenue.
Sohe’s answer was direct: the product is what ties everything together. “When you have a growth, tech, finance, and marketing team all doing separate work, the product is what unifies everyone,” he said.
His recommendation was to create a unified KPI that mirrors the product’s performance, so every team is pulling in the same direction rather than optimising for separate outcomes.
Focus on What Happens After Acquisition
Sohe closed his session with a challenge to founders on where they place their attention. “Most founders obsess over user acquisition. But what you should obsess over is what happens after the user acquisition. Where do you go from there? And what do you want to do differently?”
That shift in focus, from acquiring users to retaining and growing them, reflects the broader theme of the session: in today’s funding climate, investors are paying closer attention to companies with clear paths to profitability. Sustainable growth has become the real competitive advantage.
Cardtonic’s presence at Web Summit Vancouver and the practical lessons Sohe contributed make a strong case for why African fintech operators are increasingly relevant in global conversations about building businesses that last.
About Cardtonic
Cardtonic is a fast-growing fintech super-app enabling payments beyond borders through alternative channels such as virtual dollar cards and digital assets. The platform serves more than 1.5 million users, providing frictionless access to global payments, secure gift card exchange, international bills, and digital connectivity via eSIMs.
With a strong culture of discipline, speed, and customer trust, Cardtonic gives Africans the freedom to transact globally without relying solely on limited traditional banking rails. For more information, visit cardtonic.com.
Media Contact
Partnerships Team
clients@memoir-communications.com

President of Islamic Chamber of Commerce and Development at Global Islamic Economy Summit: World Needs Economy That Restores Ethics to Capital
Chairman of the Federation of Saudi Chambers and President of the Islamic Chamber of Commerce and Development, Abdullah Saleh Kamel, said the world needs an economic system that restores ethics to capital, warning of structural imbalances in the prevailing global economic model.
Speaking at the opening of the Third Global Islamic Economy Summit in Istanbul, Kamel said the shortcomings of the current model stem from the transformation of capital into a “negative instrument” concerned only with its owner and private gain, without regard for any adverse consequences resulting from its use, particularly for the most vulnerable groups and marginalized communities.
The summit, organized by the AlBaraka Forum for Islamic Economy under the theme “Capital in the Islamic Economy: Structuring Wealth for Sustainable Development,” is being held from June 3-6 in Istanbul under the patronage and in the presence of Turkish President Recep Tayyip Erdoğan, Sheikh Dr. Saleh bin Abdullah bin Humaid, Imam and Khateeb of the Grand Mosque, advisor at the Saudi Royal Court, member of the Council of Senior Scholars, and president of the International Islamic Fiqh Academy.
Kamel, who also serves as chairman of the Board of Trustees of the AlBaraka Forum for Islamic Economy, stressed the need for an economic system based on justice and productive spending rather than profit alone. He criticized corporate social responsibility practices in their current form, saying companies contribute only a fraction of what is needed to remedy the significant harm their practices inflict on the environment, people, and animals, in violation of the Islamic principle of preventing harm.
He noted that countries around the world are studying the introduction of legal age restrictions on social media use to protect children after studies demonstrated its harmful effects on their minds, psychological well-being, and behavior. He said this reflects the depth of the ethical imbalance in the prevailing economic model, which produces such practices and then struggles to address their consequences.
Kamel outlined three characteristics that should define capital in the Islamic economy. First, capital should be productive, generate wealth, and be spent. Second, money itself should not be traded as a commodity, which forms the basis for the prohibition of usury (riba), as it transforms money into a commodity rather than a tool serving the economy. Third, wealth should neither be hoarded nor monopolized, but developed through zakat, charitable giving, and endowments (awqaf), which constitute a form of social and charitable capital.
He said wealth concentration and rising sovereign debt are interconnected phenomena that reveal the depth of the imbalance in the prevailing global economic model. He noted that wealth continues to become increasingly concentrated in the hands of the wealthiest one percent, both within individual countries and globally, resulting in the rich becoming richer and the poor becoming poorer.
He added that the dominance of multinational corporations, particularly major technology companies, continues to expand, while governments and societies seek to reduce their negative effects.
Addressing sovereign debt, Kamel said finance ministers’ primary concern today is meeting annual debt-servicing obligations, while repayment of the principal lies beyond the scope of consideration. He said this situation places a burden on stronger economies before weaker ones and highlights deep structural imbalances in the global economic system.
Kamel attributed the root cause of these challenges to the transformation of capital into a “negative instrument” focused solely on its owner and private benefit, without consideration for the effects it leaves on others, particularly vulnerable groups and marginalized communities.
He stressed that Islamic economics offers a different structural framework for capital based on productivity, wealth creation, spending, the prohibition of trading money itself, and the development of wealth through zakat, charitable giving, and endowments. He said this framework is intended not only to benefit Muslims, but humanity as a whole.
Concluding his remarks, Kamel said the role of capital in Islamic economics extends beyond serving Muslims to benefiting all humanity. He expressed hope that the summit would serve as an influential model for Muslim capital in the service of humanity through its financial sustainability, growing impact, and leadership in shaping the future of Islamic economics.
The Third Global Islamic Economy Summit is being held with the participation of ministers, central bank governors, heads of financial institutions, and researchers from around the world.
The summit is organized by the AlBaraka Forum for Islamic Economy in strategic partnership with the Investment and Finance Office of the Presidency of the Republic of Türkiye, the Turkish Wealth Fund, the Istanbul Financial Center, Ibn Haldun University, and the Islamic Cooperation Youth Forum.
.jpeg)
Marquis Who’s Who Honors Stacey Carreiro, Who Built a Financial Career on Curiosity and Courage
Marquis Who’s Who is honoring Stacey Carreiro, senior manager of finance at Kroll LLC, for her distinguished career in the complex world of international financial operations. Ms. Carreiro approaches her responsibilities not simply as a role, but as a series of high-impact challenges requiring precision, strategic thinking and disciplined execution. Based in Oxford, New Jersey, she is focused on strengthening finance and accounting operations through a leadership style grounded in accountability, curiosity and continuous improvement.
Ms. Carreiro considers her pursuit of higher education one of her most defining accomplishments. After beginning her professional career, she made the deliberate decision to return to school in her 20s to formally pursue finance and accounting. Over the course of a demanding decade, she balanced full-time roles, career advancement and significant personal responsibilities, demonstrating resilience and sustained commitment. She earned a Bachelor of Business Administration in accounting from Kaplan University in 2013.
Continuing her academic progression, Ms. Carreiro earned a scholarship that enabled her to move directly into her graduate program, where she completed a Master of Business Administration with a concentration in finance and accounting in 2016 and was recognized on the dean’s list for her academic performance. She is currently advancing her professional credentials and is working toward certification through Payroll.org, reflecting her commitment to ongoing technical development. She also holds a Certificate in Accounting from CDM Institute and is a member of The Alpha Beta Kappa National Honor Society and the National Society of Collegiate Scholars.
A defining element of Ms. Carreiro’s career trajectory has been her long-standing professional relationship with a chief financial officer who recognized her leadership potential early on. This mentorship led to progressive roles at Jackson Hewitt, Covanta (now Reworld) and ultimately Kroll LLC, with each role expanding her scope of responsibility and operational impact.
Ms. Carreiro has consistently delivered measurable improvements across complex organizations. During her tenure at Jackson Hewitt Inc., where she spent close to a decade, her contributions were recognized with a “Best in Class” nomination, culminating in her participation in the Closing Bell ceremony at the New York Stock Exchange in 2010. Her work supported operational effectiveness across a network of more than 4,000 locations. At Covanta, she further established herself as a transformation-focused leader by streamlining accounts payable operations and advancing automation initiatives and system implementations that enhanced efficiency, scalability and control.
Through her leadership, Ms. Carreiro has emphasized accountability, process integrity and team development. She takes on challenges with energy and purpose and works effectively with teams across global regions. She believes that effective global teamwork is built on clear communication, mutual respect and an understanding of regional differences in both business practices and culture. Drawing on experience working across multiple regions, she has successfully aligned teams around shared objectives and driven consistent results in complex, multinational environments.
Looking ahead, Ms. Carreiro is strategically focused on transitioning into consulting over the next five to 10 years, where she aims to partner with a broader range of organizations to optimize financial operations and implement sustainable process improvements. Her advice to aspiring professionals reflects her own journey: “Take risks and never feel like you can't do something — because the only real limitation is choosing not to try.”
Outside of her professional accomplishments, Ms. Carreiro places a strong emphasis on maintaining balance and personal fulfillment. She enjoys traveling and values the opportunity it provides to experience different cultures, perspectives, and ways of working. In her role within a global organization, she believes that embracing diversity and working effectively with people across the world is essential to driving innovation and achieving strong business outcomes. She also enjoys reading, spending time with her family, and maintaining a busy lifestyle. Additionally, she is deeply interested in pedigree dog training and development, with a particular appreciation for the standards and practices associated with the American Kennel Club (AKC), reflecting her dedication to discipline, structure and continuous improvement both professionally and personally.
About Stacey Carreiro
Stacey Carreiro is a finance and accounting leader with extensive experience driving operational excellence across complex, multinational organizations. Currently serving as Senior Manager of Finance at Kroll LLC, she specializes in financial operations, process improvement, automation, and global team leadership. Throughout her career, Ms. Carreiro has led transformative initiatives at organizations including Jackson Hewitt, Covanta (now Reworld), and Kroll, earning recognition for her commitment to accountability, innovation, and continuous improvement.
About Marquis Who's Who
Since 1899, when A. N. Marquis printed the First Edition of Who’s Who in America®, Marquis Who’s Who® has chronicled the lives of the most accomplished individuals and innovators from every significant field, including politics, business, medicine, law, education, art, religion and entertainment. Who’s Who in America® remains an essential biographical source for thousands of researchers, journalists, librarians and executive search firms worldwide. The suite of Marquis® publications can be viewed at the official Marquis Who’s Who® website, marquiswhoswho.com.

Blaj Law Secures Final VAT Litigation Victories for Romanian HoReCa Operators
Romanian law firm Blaj Law has obtained a series of final court rulings annulling additional VAT liabilities imposed by the Romanian tax authority on HoReCa (an acronym for hotels, restaurants, and cafés/catering) operators in relation to coffee and tea-based beverages served in cafés, restaurants, bars and vending machines.
The VAT Dispute Concerning Coffee and Tea-Based Beverages
The judgments have enabled companies represented by Blaj Law to recover several million Romanian lei, and have created a relevant line of case law in a matter that affected the entire Romanian HoReCa sector. The rulings confirm that fiscal obligations must have a clear legal basis and cannot be created or extended through subsequent administrative interpretation.
The disputes concerned the VAT rate applicable to coffee and tea-based beverages following the fiscal amendments applicable from 1 January 2023. Through those amendments, certain non-alcoholic beverages were removed from the reduced VAT regime. In its considerations, the legislative change referred, in particular, to beverages containing added sugar, other sweeteners or flavourings. In the text of the law, the change targeted products falling under specific customs classification codes.
As such, the measure was generally perceived as targeting soft drinks and similar bottled products, rather than coffee or tea-based beverages prepared for immediate consumption in the HoReCa market. Coffee and tea-based beverages served in cafés, restaurants, bars or through vending machines are not typically placed on the market as bottled soft drinks and were not regarded by operators as falling under the relevant customs classification codes. They are prepared on the spot, served directly to the consumer and, in many cases, do not contain added sugar or sweeteners unless the consumer expressly requests or adds them.
For this reason, during a significant period after the entry into force of the amendments, most operators treated such beverages as continuing to benefit from the reduced 9% VAT rate applicable in Romania at the time. This approach was also supported by the fact that certain operators expressly sought clarification from local fiscal authorities as to whether the amendments applied to coffee and tea-based beverages prepared and served for immediate consumption in cafés and similar HoReCa venues. In one such case, the answer received from a local tax authority indicated that the amendment did not apply to these products. At that stage, however, no unified national interpretation had been issued.
Several months later, the national tax authority adopted a unified interpretation taking a different view, considering that certain liquid products based on coffee, tea or cocoa could fall within the category excluded from the reduced VAT regime and should therefore have been subject to the standard 19% VAT rate.
Following tax audits, operators were required to pay the 10% VAT difference for products already sold to final consumers. Since the transactions had already taken place, the prices had already been paid and the fiscal receipts had already been issued, the additional VAT could no longer be recovered from customers. As a result, the reassessed VAT became a direct cost for the businesses concerned.
For high-volume products, the financial exposure was substantial. A difference that may appear limited when assessed at the level of an individual product may have a material impact when applied across thousands or millions of transactions, and that was exactly the case here. The exposure was further increased by interest and penalties, although the taxpayers had acted in a period marked by legislative ambiguity and lack of clear administrative practice.
Court Challenges and Final Rulings Obtained by Blaj Law
Blaj Law challenged this approach before the Romanian courts on behalf of several operators in the HoReCa industry. The firm argued that the tax authority had exceeded the limits of the applicable legal framework and had unlawfully assimilated coffee and tea-based beverages to a category of products which the legislature had not intended to exclude from the reduced VAT regime.
Blaj Law further argued that, even if the wording of the legislation left room for interpretation, such ambiguity could not be resolved by adopting the most burdensome reading for taxpayers and applying it to transactions already completed. In tax matters, administrative interpretation cannot replace the law and cannot be used to extend the scope of a fiscal obligation beyond the wording, purpose and limits set by the legislature.
On June 12, 2025, the Brașov Court of Appeal issued the first final judgment in this series of disputes, upholding the annulment of the tax authority’s decisions establishing additional VAT liabilities. In that case, the court confirmed that the application of the 19% VAT rate, instead of the reduced 9% rate, to coffee and tea-based beverages was unlawful.
The same approach was followed in subsequent cases finally resolved to date, confirming that the disputed interpretation was not an isolated assessment issue, but reflected an administrative practice that could not be upheld within the limits of the applicable legal framework.
The consequences of the judgments were substantial for the firm’s clients. Once the tax claims were annulled, the taxpayers were restored to their previous legal position, and the amounts already paid became subject to restitution, together with the related interest. The rulings therefore reversed not only the legal position adopted by the tax authority, but also the economic effect of that interpretation on the companies concerned.
“Tax obligations cannot be created or extended through administrative interpretation. Where the law is ambiguous, the taxpayer cannot be penalised for adopting a reasonable and legally grounded interpretation, especially in respect of transactions already completed,” stated Bogdan Blaj, Blaj Law founder and managing partner.
Broader Implications for Taxpayers in Romania
The VAT disputes handled by Blaj Law arise in a broader fiscal context in which companies operating in Romania have faced frequent legislative amendments, increased tax burdens and inconsistent administrative practice. Romania has been undergoing an accelerated fiscal adjustment, under the pressure of major budget deficits. In recent years, the fiscal framework has been amended repeatedly, including through increases in tax rates, restrictions on preferential tax regimes, changes to reduced VAT rates, amendments to the taxation of dividends and other measures aimed at consolidating public finances and increasing budget revenues.
For companies, however, the effect goes beyond a higher tax burden. Higher taxation is a challenge that businesses may incorporate into their financial planning where the rules are clear, stable and applied consistently. A more difficult risk arises where rules are amended rapidly, their scope is unclear, secondary legislation or implementation guidance is delayed, and administrative practice varies from one period to another or from one taxpayer to another.
As an EU Member State and one of the largest markets in Central and Eastern Europe, Romania remains an important jurisdiction for cross-border investment, manufacturing, retail, energy, technology, transport and services. However, companies operating in Romania must increasingly assess not only the content of fiscal legislation, but also the risk that such legislation may later be interpreted by the tax administration in a stricter or more burdensome manner.
The central question for taxpayers is therefore not only what the law provides at a given moment, but whether the interpretation adopted in good faith at the time of the transaction will remain accepted, whether administrative practice will be applied consistently and whether the tax authority may subsequently reassess completed transactions on the basis of a different interpretation.
In this context, the final rulings obtained by Blaj Law are significant because they confirm the role of judicial review as a safeguard against unlawful or excessive administrative interpretation. Courts do not replace the legislature and do not deny the State’s right to collect taxes. However, they have the authority and obligation to verify whether a tax act was issued in compliance with the law, whether the authority’s interpretation has a valid legal basis and whether the obligation imposed on the taxpayer is lawful and justified.
The experience of Blaj Law in the VAT on coffee litigation shows that an administrative tax interpretation in Romania may be successfully challenged where it lacks sufficient legal basis. For companies, the practical conclusion is that in an unstable fiscal climate, legal defense is a necessary component of tax risk management.
About Blaj Law
Blaj Law is a Romanian law firm advising local and international clients on complex legal matters involving dispute resolution, tax and administrative litigation, corporate, commercial law and regulatory issues. The firm combines courtroom experience with strategic legal consultancy, assisting companies in preventing, managing and resolving disputes with public authorities, commercial partners and other stakeholders. Its work includes analysing and challenging tax and administrative acts, representing clients before Romanian courts, advising on regulatory and commercial risks, and managing cases in which the interpretation of legal rules may have significant economic effects on business operations.

Icarus Behavioral Health Nevada Expands its Integrated Inpatient Rehab Programs for Gambling Addiction
Icarus Behavioral Health Nevada is raising awareness of its integrated treatment services for adults facing gambling addiction alongside mental health and substance use concerns, offering structured care for a growing need in Las Vegas, across Nevada, and throughout the nation.
Gambling addiction can affect finances, relationships, employment, emotional stability, and overall well-being. For many individuals, compulsive gambling is not an isolated concern. It may occur alongside depression, anxiety, trauma, alcohol use, drug use, or other behavioral health challenges that make recovery more complex.
Icarus Nevada addresses these overlapping needs through a dual diagnosis model designed to treat the broader clinical picture, not just the outward behavior.
“Gambling addiction often carries a heavy burden of secrecy and shame, and many people try to manage it alone for far too long,” said a spokesperson for Icarus Behavioral Health Nevada. “Our goal is to help clients understand that gambling disorder is treatable, especially when care also addresses the anxiety, depression, trauma, or substance use concerns that may be present at the same time.”
With locations in Henderson and Las Vegas, Icarus Behavioral Health Nevada provides adult mental health and addiction treatment for those who need a more structured level of support. The program helps clients examine the patterns, triggers, emotional stressors, and underlying conditions that can contribute to gambling behavior while also building tools for long-term recovery.
Particularly in Las Vegas, where gaming, sports wagering, and online betting are highly visible and easily accessible, recovery can be difficult without professional support. Icarus Nevada works with clients to develop practical coping skills, relapse prevention strategies, emotional regulation tools, and healthier responses to stress and urges.
Both outpatient and residential treatment programs at Icarus include individual therapy, group therapy, behavioral health support, relapse prevention planning, and personalized care coordination based on each client’s needs. When gambling addiction occurs alongside substance use or psychiatric symptoms, the clinical team works to address both concerns together so that untreated challenges in one area do not undermine progress in another.
Icarus Behavioral Health Nevada also recognizes the importance of family education and support. Gambling addiction often affects spouses, parents, children, and loved ones who may feel confused, hurt, or unsure how to respond.
By helping families better understand compulsive gambling and dual diagnosis concerns, the program aims to reduce stigma and support more informed care decisions.
About Icarus Behavioral Health Nevada
Icarus Behavioral Health Nevada provides JCAHO-accredited, compassionate, and evidence-based care for individuals facing substance use disorders, mental health challenges, and behavioral addictions. Through personalized treatment plans, integrated inpatient programs, and a commitment to long-term recovery, the organization helps clients build healthier, more stable lives in a supportive therapeutic environment. Many forms of insurance are accepted for treatment. For more information, visit icarusbehavioralhealthnevada.com.
Disclaimer
This press release is provided for informational purposes only and does not constitute medical advice, diagnosis, treatment recommendations, or a guarantee of treatment outcomes. Individual results may vary based on a variety of factors, including personal circumstances, clinical needs, and participation in treatment. Anyone experiencing gambling-related, mental health, or substance use concerns should consult a qualified healthcare professional to determine appropriate care. References to treatment services, accreditation, or insurance acceptance are subject to change and may vary by location, provider availability, and individual eligibility.
Media Contact
Rachel Lissor
contact@icarusbhnv.com

4th Tianjin International Shipping Industry Expo Highlights AI Opportunities
The 4th Tianjin International Shipping Industry Expo (TISIE) opened in north China's Tianjin Municipality on June 2, showcasing growing AI opportunities in the shipping industry. This event is organized by Zhenwei International Exhibition Group.
Themed shipping to the world and navigating towards the future with AI leading new opportunities for the development of ports and shipping, the four-day expo covered fields such as green shipping, maritime equipment, logistics services and more.
The TISIE has been held annually in Tianjin since 2023 to promote global shipping cooperation, industry investment and trade exchange.
Xu Kai, chief information officer of Shanghai International Shipping Institute, said that China has built the world's largest network of automated container terminals, with notable breakthroughs in unmanned shore cranes, intelligent guided vehicles and automated yards.
"Terminal equipment should not only operate efficiently but also perform regional dynamic optimization based on real-time fluctuations in vessel arrivals, sudden weather changes and instantaneous cargo flow surges," Xu said. "This requires AI to evolve from executing commands to autonomous reasoning, and from single-machine intelligence to group collaboration."
Waqas Samad, CEO of Lloyd's List Intelligence, said that with the world's largest fleet, and as the world's biggest shipbuilder and producer of shipping containers, China plays a key role in today's shipping landscape. But more importantly, China represents something significant about the future of shipping, not just scale and infrastructure, but the combination of connectivity, technology and intelligence.
"AI will reshape our industry in practical and powerful ways," said Thomas Sim, president of the International Federation of Freight Forwarders Associations.
He noted that AI should empower freight forwarders, not replace their professional judgment; enhance human capability, not remove accountability; and strengthen the role of freight forwarders as trusted logistics architects, not reduce them to platform users.
Feng Boming, vice president of China Merchants Group Limited, said that AI was evolving from a conversational assistant that supports decision-making and improves efficiency to an action-oriented intelligent agent capable of autonomously understanding intentions, invoking tools and executing specific tasks.
"However, greater autonomy also entails greater security responsibilities," Feng said. "We must clearly recognize that behind AI's empowerment of thousands of industries, various new types of security risks and governance challenges continue to emerge, posing entirely new challenges to the orderly development of the industry and the safe operation of the sector."

X-VPN Completed Independent No-Logs Audit Under ISAE 3000 (Revised) Standard
X-VPN has completed an independent no-logs audit conducted by one of the Big Four auditing firms on February 28, 2026 under the ISAE 3000 (Revised) standard. The review examined whether the company’s privacy policy statements on user data handling were supported in practice by its systems, operations, and governance processes.
What the Audit Covers in Practice
The audit examined whether X-VPN’s Privacy Policy statements related to user data handling were supported in practice by the company’s systems, operational processes, and governance measures. Within that defined scope, the audit was organized around five areas tied to X-VPN’s handling of user data and the controls surrounding it. First, X-VPN does not store or record sensitive user activity information. Second, it examined the company limits data processing to the minimum user information required to provide the service. Third, VPN servers, core databases, and code are maintained in a secure and compliant manner across deployment, operation, and maintenance. Fourth, it looked at the Privacy Policy and its execution remained aligned with actual system operations and data-handling practices. Fifth, it included the mechanisms related to X-VPN’s data protection supervision framework, including those oversight processes operating with independence, transparency, and traceability.
In practical terms, this means the review was not framed around a single no-logs policy in isolation. It connected the company’s public-facing privacy commitments with the operational and governance structures that support them. The audit therefore covered both the handling of activity-related data and the broader processes intended to ensure that privacy-policy commitments are applied consistently in practice.
What the Audit Result Indicates
Based on the audit result, X-VPN does not track, collect, or store data that could identify users or link them to their online activities. This includes user IP addresses, destination IP addresses, websites visited, browsing history, VPN server information, DNS queries, downloaded content, VPN connection timestamps, and sensitive payment details. Framed this way, the outcome speaks directly to the types of records users and outside observers most often look to when assessing whether a no-logs position is meaningful in practice.
Those categories matter because they are the kinds of data that could otherwise be used to reconstruct where a user connected from, what destinations were accessed, or when activity took place. By stating the result in terms of specific data types rather than broad privacy language alone, the audit gives a more concrete foundation for understanding what the reviewed no-logs commitments cover within scope.
Industry Context: Increasing Demand for Verification
The audit comes at a time when privacy claims in the VPN market are facing closer scrutiny from both users and regulators. While “no-logs” has become a common positioning across many VPN providers, the extent to which those claims are independently examined varies widely across the industry.
In recent years, expectations have shifted from broad privacy assurances toward more verifiable forms of evidence. For users, the key question is no longer only what a provider states in its policy, but whether those statements can be assessed against actual systems and operational practices. Independent audits have increasingly become one of the ways to bridge that gap.
At the same time, regulatory attention around data handling and user privacy has continued to grow in multiple jurisdictions. This has added further emphasis on transparency, consistency between policy and practice, and the ability to demonstrate how user data is handled within defined frameworks.
Within that context, third-party assurance engagements such as no-logs audits are being used more frequently as a reference point for evaluating privacy claims. They can provide a clearer view of how a provider’s stated practices align with its operations.
Access to the Audit Report
X-VPN makes the audit report available to users through its account center. Users can access the report after logging in to their X-VPN account.
Providing access to the report matters because it gives users a direct way to review the audit outcome beyond summary statements in announcements or product messaging. In practical terms, it means the result is not presented only as a headline claim, but as a documented review that users can consult within the company’s designated access path.
Company Comment
According to Sandra Mitchell, Tech Writer and PR Lead at X-VPN, the audit is intended as part of a broader, ongoing effort rather than a one-time announcement. She described the audit as a starting point for continued transparency initiatives, including regular audits and incremental improvements to privacy and security practices.
Mitchell also stated that X-VPN plans to incorporate commonly raised concerns regarding privacy gaps and information visibility into a longer-term governance roadmap. According to her, the goal is to address these areas through trackable updates and verifiable actions rather than isolated responses.
As part of this approach, Mitchell said that X-VPN will continue publishing updates through its Transparency Report on the company's official website. She also noted that X-VPN has introduced additional privacy and security features, including post-quantum encryption and Tor over VPN, as part of its ongoing product development efforts.
In addition, Mitchell stated that X-VPN has supported organizations focused on internet security and privacy, including the Electronic Frontier Foundation (EFF) and the Internet Society (ISOC), and expects to continue supporting similar initiatives in the future.
About X-VPN
X-VPN is a global privacy and security service operated by LIGHTNINGLINK NETWORKS PTE. LTD., based in Singapore. With over 10,000 servers across 80 countries, X-VPN provides encrypted internet access using AES‑256 encryption, supporting users in protecting data, and maintaining anonymity online. The company enforces a strict no-logs policy, ensuring that no identifiable data is ever stored or shared. For more information, visit xvpn.io.
Media Contact
Sandra Mitchell
Tech Writer and PR Lead, X-VPN
sandramitchell@media.xvpn.io

SignalPlus Closes B1 Round at US$500M Valuation to Accelerate Global Expansion and Advance Derivatives Trading Technology
SignalPlus, a leading provider of institutional digital assets options and derivatives trading infrastructure, today announced the closing of its US$50M Series B1 funding round at a post-money valuation of US$500M. This round was led by HashKey Capital, with follow-on participation from BlockBooster and AppWorks. This investment reflects institutional conviction in SignalPlus’s evolution from a digital assets specialist into an institutional-grade, multi-asset trading infrastructure provider.
Goldman Sachs served as the sole financial advisor on the transaction.
Empowering Institutional Options and Derivatives Trading with Best-in-Class Technology
Founded by a team of capital markets veterans and enterprise tech builders, SignalPlus has established a reputation as the industry-leading options trading platform for institutional traders in the digital assets space. The company’s flagship trading terminal offers a Wall Street-grade position and order execution management system made accessible to the professional trader. Featuring multi-venue connectivity to the industry’s largest exchanges, it delivers unparalleled risk attribution, position analytics, and comprehensive ‘what-if’ scenario analysis exceeding the standard of current market alternatives.
Additionally, its comprehensive risk management and pricing infrastructure enables professional clients and centralized exchanges to operate automated risk management and structured product pricing around the clock. Trusted by the industry’s largest market makers and CEXs, the company achieved a record US$160 billion in platform volumes during Q4-2025, including nearly US$70 billion in Block-RFQ transactions cleared via Deribit alone. Furthermore, terminal volumes have grown at a 74% quarterly CAGR since 2023, driven by a significant rise in institutional onboarding, with the SignalPlus platform now servicing the industry’s largest options players including Cumberland, FalconX, and Galaxy Digital.
Bridging the Technology Gap Between Digital Assets and Traditional Finance
As the institutional adoption of digital assets accelerates, SignalPlus has expanded its technology suite to support structured commodity products, bringing vital modernization to a market long reliant on outdated legacy infrastructure. Proceeds from this latest fundraising round will drive product diversification and geographic expansion, building upon a trajectory that has seen the company’s market share multiply tenfold over the past 18 months.
Furthermore, SignalPlus will be releasing a comprehensive platform upgrade integrating agentic AI into its existing product workflow. Powered by the company’s proprietary QuantLab engine, users will soon be able to analyze volatility market structure, perform strategy back-testing, and create actionable trading modules — a step-change improvement over the workflow available at incumbent brokerages.
“SignalPlus was founded with the ambitious goal to be the leading infrastructure bridge connecting digital assets with traditional capital markets, a journey defined by strict operational discipline at every cycle. This funding round powers the next inflection point - growing our crypto leadership against a maturing regulatory backdrop, extending our institutional-grade infrastructure into traditional finance, and launching the SignalPlus 2.0 platform to bring agentic AI capabilities into a secure trading environment accessible to every user," said Chris Yu, co-founder and CEO of SignalPlus.
“The digital asset market is entering a new phase of institutional maturity, and SignalPlus has established itself as the leading infrastructure provider in the crypto options space, delivering a platform that meets the highest standards of operational rigor, risk management, and liquidity demanded by top-tier institutions globally. We believe the next wave of digital finance will win at the infrastructure layer, and in options, SignalPlus is already defining that standard. As regulatory clarity accelerates across key markets, this partnership will be central to expanding Hashkey Group's product offerings and reinforcing our leadership across the institutional digital asset ecosystem. We are pleased to lead this round and to deepen our long-term partnership with the team,” said Deng Chao, CEO of HashKey Capital.
"SignalPlus provides the infrastructure underpinning institutional participation in digital asset derivatives, combining the liquidity, risk management, and operational rigor that sophisticated players demand. Our relationship with the team long predates this round, and this investment directly aligns with a core thesis BlockBooster has long held: that institutional capital will enter digital assets through credible, enterprise-grade infrastructure. As a full-stack alternative asset manager pairing an incubation engine with capital management, we see SignalPlus sitting precisely at that intersection. We anticipate deep collaboration as their infrastructure connects with our capital strategies and broader ecosystem, and we are proud to back that vision with a significant commitment as the team enters its next phase of growth," said Samuel Gu, founder and CEO of BlockBooster.
About SignalPlus
Headquartered in Hong Kong, SignalPlus builds institutional-grade derivatives trading infrastructure for the converging capital markets. Its platform provides professional options analytics, real-time risk management, and execution tools to hedge funds, market makers, proprietary trading desks, and asset managers across digital and traditional financial markets. The company partners with the industry’s leading exchanges and trading institutions, and is backed by HashKey Capital, AppWorks, Tencent, and other prominent technology and financial investors.
Media Contact
Corri Wang
pr@signalplus.com

Campfire Named to Inc.’s 2026 Best Workplaces List
Campfire is proud to announce it has been named to Inc.’s 2026 Best Workplaces list. The list honors American companies that have built exceptional workplaces and vibrant cultures that support their teams and businesses, whether in-person, remote, or hybrid.
The award is the result of comprehensive measurement and evaluation of hundreds of applicants. The process involved a detailed employee survey conducted by Quantum Workplace, covering critical elements such as management effectiveness, perks, professional development, and overall company culture. Each company’s benefits were also audited to determine the overall score. Campfire is honored to be included among the 507 companies recognized this year.
"This recognition means a great deal because it comes directly from the people who make Campfire what it is," said John Glasgow, founder and CEO of Campfire. "We've grown from 10 to over 115-plus employees over the last year by hiring people with exceptional drive, curiosity, and ownership. We believe the best talent is looking for growth, and we've built a culture where people can learn quickly, take on real responsibility, and have a meaningful impact from day one.”
Campfire builds AI-native ERP software for modern finance and accounting teams. Designed as a full replacement for legacy systems, Campfire brings together the general ledger, revenue automation, close management, and reporting in one unified platform. Its proprietary AI technologies, including its Ember agents, are trained exclusively on accounting data to automate tasks such as reconciliations, anomaly detection, and report drafting with industry-leading accuracy. This allows finance teams to close faster, reduce manual work, and focus on analysis instead of administration.
“This year’s Best Workplaces list goes beyond great company culture — it highlights companies making meaningful and sustained investment in their employees,” said Bonny Ghosh, editorial director at Inc. “Even in a labor market that favors employers, these companies understand that an intentional and authentic commitment to their teams drives stronger employee retention, engagement, and ultimately, a stronger business overall.”
To view the full list of winners, visit www.inc.com/best-workplaces/2026.
About Campfire
Campfire is the AI-native ERP for high-growth companies. They give modern, mid-market and enterprise accounting teams superpowers by automating the work that nobody wants to do: manual transaction categorization, bank reconciliation, revenue recognition, and variance analysis. Their customers close 5x faster and save up to hundreds of thousands annually. Campfire is privileged to work with some of the fastest-growing AI companies in the world, including Replit, Decagon, and PostHog, as well as services providers, health tech, and aerospace companies. For more information, visit campfire.ai.
About Inc.
Inc. is the leading media brand and playbook for entrepreneurs and business. Through its journalism, Inc. aims to inform, educate, and elevate the profile of its community: the risk-takers, the innovators, and the ultra-driven go-getters who are creating the future of business. Inc. is published by Mansueto Ventures LLC, along with fellow leading business publication Fast Company. For more information, visit www.inc.com.
About Quantum Workplace
Quantum Workplace, based in Omaha, Nebraska, is an HR technology company that serves organizations through employee-engagement surveys, action-planning tools, exit surveys, peer-to-peer recognition, performance evaluations, goal tracking, and leadership assessment. For more information, visit www.quantumworkplace.com.
Media Contact
Katrina Queirolo
katrina@campfire.ai

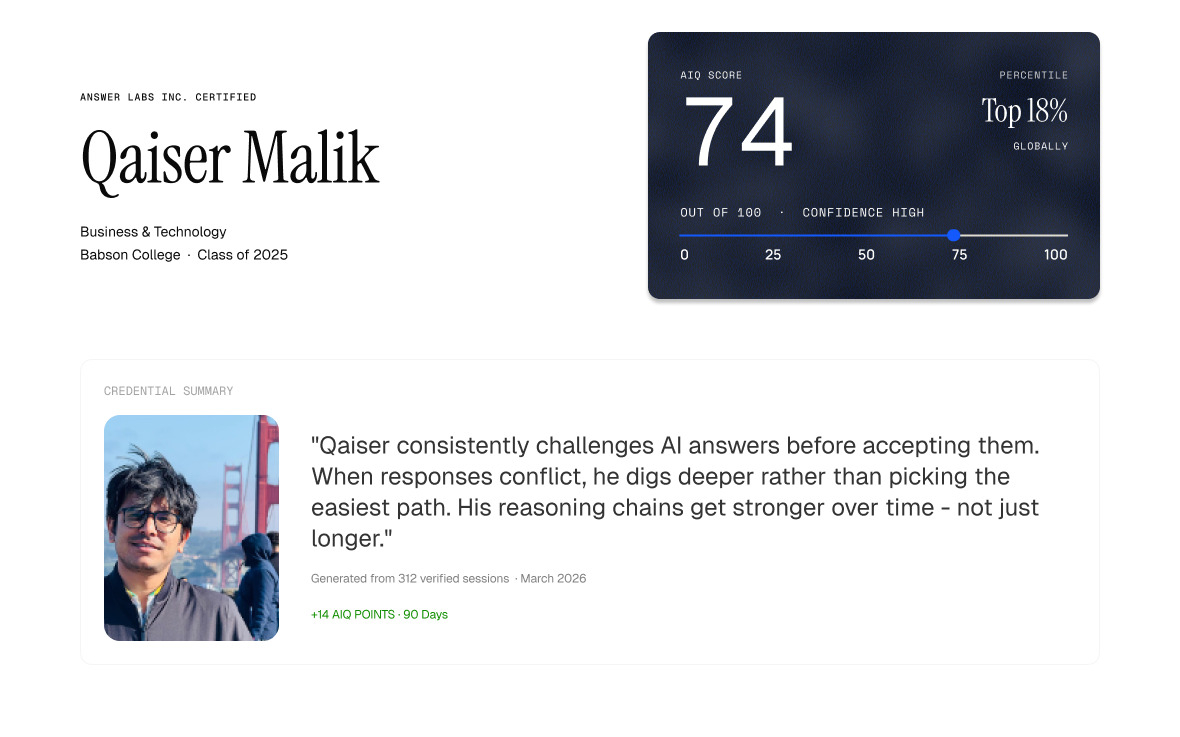
Answer Labs Launches AIQ to Verify Human Judgment in AI-Assisted Work
Generative AI has made finished work easier to produce and harder to interpret. Two people can submit equally polished AI-assisted work while demonstrating very different reasoning. Most institutions still see only the output. For more than a century, institutions evaluated people through exams, grades, resumes and credentials. In the age of generative AI, output alone is no longer enough to measure reasoning, judgment, or understanding.
Today, Answer Labs Inc., the company behind the Answerr platform, launched AIQ™ — a learning-provenance system that documents how a person reasons with AI across a body of work and converts that behavioral evidence into a verifiable AIQ profile and credential. The company describes AIQ as “proof of intelligence for the age of AI.”
Rather than detecting whether AI was used, AIQ evaluates how it was used. The system analyzes behaviors such as verifying claims, refining prompts, challenging outputs, connecting sources and recovering from weak AI responses.
AIQ is built for universities, employers and learning platforms that need to evaluate AI-assisted work without banning tools or relying on AI detection. As AI fluency becomes increasingly important in admissions, hiring and workforce development, AIQ is designed to provide institutions with a standardized signal of AI-assisted reasoning and judgment.
“AI can generate infinite content. Human judgment is becoming a scarce resource,” said Mohd Qaiser Malik, co-founder and CEO of Answer Labs Inc. “The future will not reward people for accessing AI. It will reward those who think well while working alongside it.”
AIQ is built on SLPT, the Standard Learning Provenance Taxonomy — an open specification for structuring behavioral evidence in AI-mediated learning environments.
The reference implementation includes 178 tests and five interoperability adapters — LTI 1.3, xAPI, CLR 2.0, cmi5, and Common Cartridge — enabling compatibility with systems institutions already use.
SLPT has been published under the Apache 2.0 license with a registered DOI (10.5281/zenodo.20297510), allowing institutions, researchers, and developers to inspect, validate, and build on the framework openly.
The framework was developed with contributions from Trond Arne Undheim, author of "The Platinum Workforce: How to Train and Hire for the 21st Century’s Industrial Transition," and Nada Hashmi, co-founder of Answer Labs and MIT Research Affiliate.
Answer Labs is also part of the MIT Startup Exchange (STEX), a corporate-startup collaboration platform, and is currently inviting universities, enterprises, LMS providers and workforce organizations to collaborate on the next phase of AI governance and learning-provenance infrastructure.
About Answer Labs Inc. (Answerr)
Answer Labs Inc. is an AI infrastructure company building systems for trusted learning, human judgment verification, and AI-assisted education and workforce environments. Its flagship platform, Answerr, provides institutions and enterprises with infrastructure for AI governance, learning provenance and AI-mediated decision-making. For more information, visit answerr.ai.
Media Contact
Mohd Qaiser Malik
qaiser@answerr.ai
PatSnap Releases 2026 R&D Benchmark Report, Revealing the Intelligence Gap Costing Innovation Teams Millions
PatSnap, the AI-native innovation intelligence company for IP and R&D teams, today released its "2026 R&D Benchmark Report: Waste, AI and the Race to Market." The report draws on responses from more than 200 senior R&D professionals across North America, the U.K., and Europe, spanning nine industries, all with annual R&D budgets of at least $50 million.
PatSnap commissioned the research to quantify what its customers have long described: that even well-resourced, AI-enabled R&D organizations are making consequential decisions on incomplete intelligence, and paying for it. The findings confirm that the problem is structural, measurable, and addressable.
"We commissioned the '2026 R&D Benchmark Report' because our customers kept describing the same problem: their AI was capable, but the intelligence feeding it was not keeping pace. We wanted to know whether that was isolated frustration or a reality across the market,” said Jason Resnick, General Manager, PatSnap West. “The data confirmed it was consistent across every industry and budget level we surveyed. Patent landscapes shift. Competitive pipelines move. Intelligence that was accurate last quarter can send a team in the wrong direction. In R&D, the cost of acting on incomplete intelligence is not a delayed report. It is months of work that cannot be recovered."
Key findings include:
- R&D waste is significant and recurring: More than a third of organizations (37%) spend between 25% and 40% of their entire R&D budget on projects that never reach the market. When projects are terminated late in development or testing, 45% of respondents estimate the wasted investment at $1 million or more per project.
- Most projects don't make it: Nearly three quarters of respondents (73%) report that half or fewer of their initiated R&D projects ultimately reach market launch.
- AI adoption is near-universal, but the intelligence gap persists: 92% of respondents are using AI in their R&D process at least moderately, and 70% say implementations have been largely successful. Among the heaviest AI users, 54% say better, faster access to intelligence would still have the greatest impact on their R&D success, a higher rate than among moderate users (43%).
Alongside the report, PatSnap is launching an interactive R&D Benchmark Calculator that allows organizations to compare their own spending and project outcomes directly against survey respondents. Users can input details including their industry, annual R&D spend, the share of projects reaching market, average development cycle length, and typical project cost, to understand how their organization stacks up against peers across the survey.
Download the report: "2026 R&D Benchmark Report: Waste, AI and the Race to Market"
About PatSnap
Founded in Singapore in 2007, PatSnap is a leading global innovation intelligence company. Leveraging domain-specific AI agents and robust analytics tools, PatSnap empowers IP and R&D teams to unlock insights, accelerate discoveries and protect breakthroughs. Trusted by nearly 20,000 enterprises, law firms and research institutions in over 50 countries including one-third of the world’s largest R&D spenders, PatSnap transforms how organizations innovate and make critical decisions. Learn more at www.patsnap.com.
Media Contact
Kate White
media@patsnap.com

WorkWise Robotics Named Workforce Partner for Autonomous Deployment at PortMiami, Supporting Smart City Infrastructure
At eMerge Americas 2026, Miami-Dade County named Miami-based WorkWise Robotics as the workforce partner behind an autonomous deployment at PortMiami — a future-ready bet designed to expand operational capacity, elevate the passenger experience, and reposition the existing facilities team into higher-value roles before labor pressures force less-considered choices.
As public infrastructure demand outpaces workforce capacity across U.S. cities, the deployment marks a shift in what smart city investment looks like, turning the gap into an opportunity. The digital layer — dashboards, connectivity, agentic AI assistants — has dominated the conversation for a decade. The physical layer, the workforce that actually maintains terminals, concourses, and public-facing facilities, has largely been treated as a given.
PortMiami, the Cruise Capital of the World® and Cargo Gateway of the Americas®, is now the proving ground for what a smart city strategy looks like when both layers move forward together.
“This is what the future looks like — and it’s being built right here in Miami-Dade,” Mayor Daniella Levine Cava said at the unveiling. “We’re not just keeping pace with innovation. We’re leading it, harnessing artificial intelligence to transform how we serve our visitors and our community every day. From our airport to our seaport, we are creating a smarter, more connected, and truly Future-Ready Miami-Dade.”
A Robotics Workforce at the Country’s Busiest Cruise Port
WorkWise Robotics deployed autonomous cleaning robots at PortMiami during peak cruise season, when the terminal processes its highest volume of passengers per day and the operational tolerance for downtime is lowest. The robots operate alongside the maintenance team, providing continuous floor coverage across high-traffic terminal areas — freeing staff to focus on specialized cleaning tasks where human judgment and detail matter most.
“The innovation from WorkWise Robotics is supporting our maintenance and cleaning staff by enabling them to focus on more technical and specialized cleaning tasks,” said Gustavo Grande, Head of Innovation at PortMiami. The biggest return, Grande noted, is felt “during passenger days and peak times, when the robots manage the heavy load of cleaning while staff focus on bathrooms and other high-touch areas to maintain an overall high-hygiene environment.”
The deployment converts what had been a coverage problem — too much demand for too few staff-hours — into a workforce evolution. Cleaning consistency improves because the robots run continuous shifts across high-traffic floors. Specialized cleaning quality improves because the maintenance team is freed for the technical work where their judgment matters. The passenger experience improves because both happen simultaneously, rather than sequentially.
Smart City Innovation, Rethought
The hardest lesson of the last decade of smart city investment is that dashboards don’t maintain buildings. Floors still need cleaning. Terminals still need turnover. Passengers still notice. Miami-Dade’s Future-Ready strategy invests across both layers — digital traveler-facing tools at Miami International Airport, and WorkWise’s robotic deployment at PortMiami.
“The digital layer is getting smarter. The physical layer is getting thinner,” said Cam Parra, Chief Executive Officer of WorkWise Robotics. “PortMiami is proving that the next layer of smart city investment isn’t another dashboard. It’s a robotics workforce that keeps the physical environment performing at the same level as the digital one. Ports and public infrastructure facilities around the world face the same structural challenge — aging maintenance models, overextended workforces, rising demand. The cities that figure out how to modernize the physical layer first will set the standard. That’s what we’re doing in partnership with Miami-Dade and PortMiami, and it’s paying off in operational efficiency and in passenger experience.”
The Workforce Question Smart Cities Can No Longer Defer
South Florida’s public infrastructure is in the middle of an unprecedented capital expansion. Miami International Airport is investing $14 billion in capital improvements through the end of the decade. PortMiami contributes $43 billion in annual economic impact and supports more than 334,000 jobs, with cruise lines committing additional terminal investments through 2027. New gates. New terminals. New concourses. And, year over year, more passengers.
The workforce maintaining that infrastructure is not scaling at the same pace. Building and grounds maintenance employment, according to the Bureau of Labor Statistics, is projected to grow well below the rate of demand through the end of the decade. In tourism-driven economies, the resulting capacity gap shows up directly in the visitor experience: floors, restrooms, terminals, concourses — the spaces where passenger perception is formed.
For airport and seaport leaders, this is not just a capacity question — it’s a workforce evolution question. The window to reposition the existing facilities team into higher-value roles is open now, while operational pressure is still manageable. Counties that wait until the labor gap forces the issue end up backfilling rather than upskilling, and the institutional knowledge built over years of service walks out the door with the people who held it. It is also a stewardship question: every dollar of capital improvement funds a facility that must be maintained, every additional passenger raises the bar on cleaning and turnover, and every workforce gap left unaddressed shows up in the visitor experience that drives convention bids, cruise bookings, and regional economic competitiveness. A smart city investment that does not evolve the workforce is an investment that does not protect the asset — or the people maintaining it.
A robotic workforce solves four things at once: it opens an upskilling pathway for the existing facilities team into higher-value roles — robot fleet coordinators, performance analysts, hybrid workforce supervisors; it elevates the passenger experience through continuous coverage of high-traffic spaces; it expands operational capacity in step with rising demand; and it extends what the existing team can deliver without expanding the public payroll. The workforce evolves rather than stretches. The visitor experience improves. The same taxpayer dollar goes further.
Under the Robots-as-a-Service model, the effective cost is roughly 30 cents per robot operating hour — designed to fit operating budgets, not capital approval cycles.
A Workforce That Evolves
That four-part promise — upskilling, experience, capacity, payroll efficiency — is only as good as the deployment model that delivers it. WorkWise’s deployment model has one rule: no robot goes live before the people on the ground are trained to manage it. Existing facility staff are trained through WorkWise Academy to operate the robots, interpret performance data, and coordinate fleets across shifts.
“Our mission is empowering the next generation of work — where facilities workers become robot fleet coordinators,” Parra said. “Every deployment starts with the people already on the ground. Before a single robot goes live, the existing team goes through WorkWise Academy. They learn to operate the machines, read the performance data, and coordinate a hybrid workforce. By the time we hand over, the team isn’t watching the robots — they’re managing them. That’s the difference between deployments that disrupt and deployments that elevate.”
WorkWise operates on a Robots-as-a-Service (RaaS) model — a monthly subscription that includes hardware, software, maintenance, and ongoing customer success support. The model is purpose-built for institutional and enterprise buyers who need to validate measurable outcomes before committing to a long-term roll out.
“We start with the single workflow consuming the most labor hours with the least return,” Parra said. “Deploy a targeted robotic solution, measure everything for 30 days, and let the data make the case. The pilot generates evidence before it requires commitment — critical when you’re talking about public facilities or large commercial operations.”
The Wave Is Here
The shift to a robotic public infrastructure workforce is no longer something city officials can wait out. Tampa International Airport, Salt Lake City International Airport, and Pittsburgh International Airport all introduced autonomous cleaning fleets during the pandemic and never sent them back. By 2024, nearly 100 autonomous cleaning robots deployed across airport environments had logged almost 10,000 operating hours and cleaned more than 35 million square feet of terminal space. The operational case is settled.
What’s now emerging is the strategic conversation — and Miami-Dade is leading it in public. By unveiling WorkWise’s deployment at eMerge Americas 2026, the County’s flagship innovation event, Miami-Dade did more than disclose a vendor partnership. It positioned a robotic workforce alongside its other Future-Ready announcements, signaling that physical-layer infrastructure investment now sits at the same strategic altitude as digital-layer investment. Miami International Airport CEO Ralph Cutie has publicly tied the airport’s $14 billion modernization plan to a vision of global competitiveness anchored by both physical and digital innovation.
For mayors, port directors, and aviation leaders watching the pattern, the playbook is becoming clear: identify the workflows where labor pressure is highest and visitor visibility is greatest, deploy a targeted robotic solution, measure for 30 days, and let operational data and workforce outcomes guide expansion. The cities that move while the upskilling window is open will set the standard the rest will spend the next decade trying to replicate.
What Comes Next
WorkWise is in active discussions with public infrastructure operators across Florida and evaluating expansion into other major U.S. metro markets facing similar workforce capacity pressures.
“WorkWise brings together a best-in-class robotics ecosystem and a customer success model that ensures robots and humans work collaboratively to amplify impact and scale,” Parra said. “That’s what makes our model fit the public institutions, like Miami-Dade, where workforce evolution and operational excellence have to advance together. For the cities watching this trend, the window to lead is open.”
About WorkWise Robotics
WorkWise Robotics is a Miami-based robotic workforce company deploying autonomous cleaning, cargo transport, and service robots through a Robots-as-a-Service model to hospitality, transportation, cruise, retail, public infrastructure, and warehousing enterprises across the United States. Founded by operators from McKinsey, BCG, Google, and Microsoft, WorkWise pairs commercial robotics with the enterprise transformation discipline required to institutionalize adoption — sequencing rollout, aligning executive sponsors, training existing teams through WorkWise Academy, and measuring outcomes that move the business. Learn more at workwiserobotics.com.
Media Contact
WorkWise Robotics
press@workwiserobotics.com

Enterprise Robotics Has Reached Its Inflection Point: WorkWise Robotics CEO Cam Parra on Why the Companies Moving Now Will Define the Next Decade of Customer Experience
Service-sector demand is climbing faster than workforce capacity for the first time in a generation. The lesson from enterprise AI adoption is what happens next: AI-native startups built market positions in less than two years that incumbent SaaS players had spent a decade building — and forced those incumbents into reactive product cycles they’re still trying to escape. The companies that moved early on AI integration built moats. The ones that treated it as a watch-and-wait technology lost market share to entrants that didn’t exist five years ago.
Robotics is now positioned to do to service businesses what AI did to software. The enterprises building a robotic workforce into their operating model in 2026 are constructing the same kind of moat — and the companies that wait will spend the next decade competing against operators who used those years to deliver customer experiences they cannot match.
And across hospitality, transportation, retail, cruise, and public infrastructure, enterprise leaders have started institutionalizing — turning autonomous service robots into a permanent layer of their workforce strategy. Cam Parra, CEO of WorkWise Robotics — the Miami-based company behind more than 90,000 commercial robot deployments worldwide — sees the inflection point as a true turn of the page onto the next decade of competitive advantage for service players.
The evidence is now visible across every service-driven sector:
- Airports & Ports: Autonomous cleaning fleets have moved from pandemic-era pilots to permanent operations at major U.S. airports and seaports — improving cleaning consistency, lifting passenger satisfaction scores, and freeing maintenance teams for specialized work that requires human judgment. Autonomous cargo and baggage transport robots are accelerating dock-to-terminal flows and reducing the lost-luggage incidents that drive compensation expense and CSAT decline.
- Retail: Big-box operators are deploying mobile retail-media units — moving promotional shelf space and digital ad displays into high-traffic zones — to lift same-store sales, increase basket size, and create a new monetizable surface for supplier-funded campaigns. Restocking robots are moving cases and totes across sales floors to accelerate dock-to-pick cycles and improve on-shelf availability, two of the highest-leverage levers in retail OKRs.
- Hotels & Cruises: Cleaning robots are taking on the heavy load of large-area floor care — lobbies, corridors, hallways, dining floors, decks — so cabin and room attendants can concentrate on the cabin and room turnover speed that determines whether the next guest checks in to a ready space on time. Food-runner and bussing robots are absorbing the repetitive transport work that consumed front-of-house labor hours during peak service, redirecting that time into guest interaction, upselling, and the touchpoints that drive repeat visits and brand loyalty.
- Public infrastructure: Municipalities are deploying robotic cleaning and service workflows in public-facing facilities — ports, transit hubs, civic spaces — to expand operational capacity ahead of rising passenger volumes, with Miami-Dade County’s deployment at PortMiami unveiled at eMerge Americas 2026 emerging as the public-sector reference case.
- Warehousing and logistics: Inventory management and autonomous transport robots are improving stock accuracy, compressing cycle counts, and lifting order-fulfillment speed during the peak seasons that determine whether operators meet annual revenue targets.
The common thread across every vertical is the same: robots absorbing the high-volume repetitive work that doesn’t differentiate the brand, frontline teams concentrating on the work that does.
“The companies investing today see payback inside ninety days, and they’re positioning to play offense when the next demand cycle hits,” Parra said. “The ones waiting will spend the same capital playing defense — catching up to competitors who already moved. The math favors those who move early.”
Two paths from the same starting line
Every CEO in a service-driven industry is standing at the same inflection point. From that point, two paths run in opposite directions — and they widen with every quarter that passes.
On one path, operators integrate a robotic workforce into the workflows where it produces the highest leverage — floor care, food and bev transport, internal logistics, restocking, room delivery. Their frontline teams are redirected from repetitive physical work into the moments that build brand: personalization, problem-solving, anticipating guest needs, curating experiences. Robotics-generated capacity gets reinvested into the customer experience layer that drives repeat business, loyalty, and lifetime value.
On the other path, operators wait — watching pilots, requesting more data, deferring procurement decisions. Their frontline teams stay stretched across the same workload that was already overextended in 2024. Customer experience slips not in any visible way, but in the small moments that compound into satisfaction scores, repeat bookings, and brand preference. Twelve to eighteen months in, the gap on the competitor running the other path becomes operationally and financially material.
“The cost of inaction is simply losing ground on customer lifetime value, retention, and loyalty while competitors invest in better experiences without impacting margin,” Parra said. “Every hour their frontline team spends on guest experience instead of repetitive tasks widens the gap. That asymmetry compounds.”
Why the moat compounds — and the late mover pays twice
The economics of early adoption aren’t linear — they compound. A hospitality operator that deploys robotic floor care and back-of-house transport in 2026 doesn’t just save labor hours that quarter. They reinvest those hours into guest-facing service quality, which lifts satisfaction scores, which lifts repeat-booking rates, which lifts revenue per available room over the next three to four cycles. The capital that funded the original robotics program is recovered inside ninety days and then continues generating return through the customer experience flywheel it set in motion.
The operator on the other path doesn’t just stand still. They lose share to the competitor who moved first — because guest expectations don’t reset. Once a traveler experiences the cleaner property, the faster room service, the more attentive concierge, the more responsive cabin crew, the baseline they hold their next booking to has moved. The late mover now faces two costs simultaneously: catching up on the operational capability they didn’t build, and rebuilding loyalty with guests who have already chosen a different brand.
“WorkWise’s mission is to empower the next generation of work — where robots and humans work collaboratively to amplify impact and scale,” Parra said. “The companies pulling ahead aren’t replacing teams. They’re equipping them to deliver experiences their competitors can’t match.”
What playing offense actually requires
The same enterprises that are now setting the pace didn’t arrive there by buying robots. They arrived there by treating robotics adoption as a workforce strategy with a multi-year horizon — sequenced carefully, integrated with existing operational rhythms, and measured against business outcomes, not pilot metrics.
“Robotics is a long-duration strategy, not a tactical purchase,” Parra said. “The companies that move now are scaling their workforce capacity ahead of the next demand cycle — letting robots absorb the eighty percent of repetitive work that doesn’t differentiate them, so their people can focus on the twenty percent that builds brand loyalty and the experiences guests remember.”
The strategic premise behind that quote is the operational principle that separates the offense path from the defense path. Robotics doesn’t replace the workforce; it concentrates the workforce on the work that drives competitive differentiation. Floor care, food running, supply transport, restocking — the work that gets done either way and that no guest remembers — moves to autonomous systems. Hospitality, judgment, problem-solving, brand delivery — the work that determines whether a guest returns — moves to the front of every frontline team’s day.
The partner question
“The mistake I see is treating robotics like a technology purchase. It isn’t. It’s a workforce strategy that touches every part of how a service business operates — from how frontline teams are trained, to how leaders measure performance, to how the brand promise is delivered every shift. The companies that get this right design the strategy first and pick the technology second. That’s where WorkWise comes in — as the technology and operational consulting partner that helps enterprises design the strategy, sequence the rollout, and measure the outcomes,” Parra said.
A robotics program isn’t a procurement decision — it’s a workforce strategy. WorkWise was built on that insight, by operators from McKinsey, BCG, Google, and Microsoft who have led the kind of enterprise transformation programs that move thousands of people through new ways of working. They bring the discipline of designing SOPs and OKRs, leading change management at scale, aligning executive sponsors, and measuring outcomes that move the business. That’s where technology meets operational consulting at its finest — and where most robotics adoption programs stall without it.
WorkWise operates on a Robots-as-a-Service model that aligns to operating budgets rather than capital approval cycles. Existing facility staff are trained through WorkWise Academy to operate the robots, interpret performance data, and coordinate fleets across shifts — turning a procurement event into a workforce evolution. The same playbook that institutionalizes the technology institutionalizes the people running it.
The decision in front of every service-sector CEO
The window to lead this curve, rather than chase it, is open right now. Adoption signals across hospitality, transportation, cruise, and retail are converging on a single conclusion: the operators who institutionalize a robotic workforce in 2026 will define their industry’s customer experience benchmark for the rest of the decade. The operators who wait will be reacting to that benchmark, not setting it.
“The strategic question isn’t whether robotics will be adopted as a workforce strategy. It’s at what scale, and across what departments first,” Parra said. “The companies that move first will set the pace for their industry. The ones that wait will be running someone else’s playbook.”
About WorkWise Robotics
WorkWise Robotics is a Miami-based robotic workforce company deploying autonomous cleaning, cargo transport, and service robots through a Robots-as-a-Service model to hospitality, transportation, cruise, retail, public infrastructure, and warehousing enterprises across the United States. Founded by operators from McKinsey, BCG, Google, and Microsoft, WorkWise pairs commercial robotics with the enterprise transformation discipline required to institutionalize adoption — sequencing rollout, aligning executive sponsors, training existing teams through WorkWise Academy, and measuring outcomes that move the business. Learn more at workwiserobotics.com.
Media Contact
WorkWise Robotics
press@workwiserobotics.com

Brosix Marks 20 Years in Team Messaging
Brosix, a private team messaging platform headquartered in Wilmington, Delaware, is marking its 20th anniversary this month, having first published the platform in June 2006. To commemorate the milestone, the company is offering new annual plan subscribers 15 months of service for the price of 12 — three bonus months — for plans started by June 15, 2026. The anniversary comes as Brosix continues to serve small and mid-sized businesses seeking team communication tools that balance ease of use with administrative control.
Brosix was founded in 2006 to address a gap that founder Stefan Chekanov observed firsthand: consumer chat applications were convenient but lacked the security, structure, and oversight that businesses depend on, while the enterprise-grade alternatives of the era required on-site servers, dedicated IT staff, and significant hardware investment that placed them out of reach for most small and mid-sized teams.
"We started Brosix because we couldn't find anything in the middle — something easy enough that employees would actually use it, but built properly for a business," said Stefan Chekanov, founder and President of Brosix.
"Twenty years later, that gap is still real. Small and mid-size teams don't need a corporate platform designed for organizations many times their size. They need communication that just works."
The company says the core principles behind the platform have remained consistent since launch: straightforward daily use for employees, fast setup and management for administrators, and built-in security and control that teams do not have to configure themselves.
The platform today supports one-on-one and group messaging, voice and video calls, screen sharing, remote desktop access, and secure file transfer across Windows, macOS, Linux, iOS, Android, and the web.
In a review posted to Capterra in April 2026, a Director of Vendor Management in the insurance industry, wrote:
"We have been using Brosix as our primary internal instant messaging platform since around 2012, and it has been an outstanding part of our daily operations ever since. Brosix excels at what an instant messenger should do: fast, dependable, distraction-free communication."
The company cited the review as representative of the long-term relationships it has built with customers over two decades.
Chekanov noted that the landscape has shifted considerably since 2006 — remote and distributed work is now common across industries and company sizes — but said the fundamental need Brosix was built to address has not changed. The company's 20th-anniversary promotion runs through June 15, 2026, and is available to any new annual plan subscriber.
About Brosix
Brosix is a private team messaging platform built for small and mid-sized businesses. Founded in 2006 and headquartered in Wilmington, Delaware, Brosix combines the simplicity of consumer chat applications with the security, control, and administrative oversight that companies require. The platform supports one-on-one and group messaging, voice and video calls, screen sharing, remote desktop access, and secure file transfer across Windows, macOS, Linux, iOS, Android, and the web. Brosix gives teams a private communication network with no third-party access, no ads, and no distractions. Learn more at www.brosix.com
Media Contact
Stefan Chekanov
pr@brosix.com

AVIDLOVE Debuts at PARAISO Miami Swim Week with 'Sparks Fly'
In May 2026, emerging brand AVIDLOVE made its waterfront runway debut at the 22nd Miami Swim Week, presenting "Sparks Fly with AVIDLOVE" at the RISE show venue. The brand wove lingerie elements into resort settings, injecting bold, laid-back energy into Swim Week.
Runway on Water: Where Sensuality Meets Freedom
Above the pools of Miami Beach, models showcased lace, mesh, and cut-out pieces set against a backdrop of live DJ sets, clean-girl makeup, and editorial hair — a visual spectacle of literal sparks flying. From lace camisoles with denim shorts poolside, to mesh tops with high-waisted skirts at sunset parties, to sheer two-piece sets for beach date nights, every look was designed to transition effortlessly into real vacation life.
Trend Decoded: From "Lingerie" to "Island Essential"
AVIDLOVE's RISE show signaled three key trend directions for the season:
- Lace + Mesh: From the Bedroom to the Beach
Lace camisoles and fishnet tops shed their private connotations to become core island wardrobe pieces, layered with hot pants or cover-ups for added dimension. - Y2K Revival: Millennial Style Becomes the New Resort Favorite
Slip dresses, low-rise skirts, and lace crop tops — signature Y2K elements — reimagined in bright, soft tones to evoke a youthful, dynamic vacation mood. - One Piece, Many Ways: The Year-Round "Travel Capsule"
A camisole top works as both a base layer and a standalone piece. A mini skirt can be worn solo or layered. A mesh top doubles as a bikini cover-up or pairs with trousers for street style — truly "one garment, multiple scenarios."
Runway-to-Real-Life Looks
To bring the "innerwear-as-outerwear" concept to life, AVIDLOVE launched runway-identical styling options:
- Cut-Out Crisscross Mesh Tank: A chest-crossing strap design creates an instant focal point. The high-stretch, breathable fabric flatters a range of body types — wear it with jeans for the beach or under an unbuttoned shirt for the street.
- Y2K Lace-Up Lace Crop Top: Delicate lace meets a front tie-closure in a flattering short silhouette. Doubles as a bikini cover-up or pairs with a midi skirt for a sweet-meets-edgy resort look.
- V-Neck Mesh Camisole Two-Piece: A ruched cami top with a low-rise mini skirt. The set does the styling for you — effortless for beach parties or brunch alike.
- Lace Baby Doll Dress: Sheer lace with a matching garter belt. Wear it privately or layer it under a denim jacket for a contrast-driven, spicy-girl outfit.
- Lace Camisole Top: Lightweight and sheer, this piece works tucked under a shirt or worn solo on vacation — a true multi-scene staple.
From Miami to the World: AVIDLOVE's "Sparks" Are Spreading
When lace no longer hides at the back of the closet, when mesh becomes a sight under the sun — AVIDLOVE used its "Sparks Fly" show above the Miami pools to redefine the boundary between lingerie and resort wear. This is more than a fashion moment; it is the brand's emotional message to every woman: lingerie can be worn out loud, worn daily, worn confidently.
From Miami to the globe, AVIDLOVE is igniting women's imagination around freedom, sensuality, and effortless styling. Runway-identical pieces are now available on the official flagship store, with influencer collabs bringing the show looks to life. Explore the collection and craft your own "innerwear-as-outerwear" signature.
For more information, please visit the AVIDLOVE website and Amazon storefront, or connect with AVIDLOVE on Facebook and Instagram.

symmetRE Becomes the AI Foundation for Real Estate Asset Management
symmetRE, the reporting and intelligence platform for real estate owners and operators, today announced the general availability of its Model Context Protocol (MCP) connector — the first release on the company's shared AI foundation, and the opening move in a broader strategy to continue to stay at the edge of innovation and be a best-in-class AI partner for CRE owners and operators.
The MCP connector lets symmetRE clients access their full portfolio data — every dashboard and widget they already have permission to view — from inside any AI chat interface that supports the Model Context Protocol, including Claude, ChatGPT, and Gemini. Setup is one-time; from launch day forward, owners and asset managers can ask portfolio questions in the AI tools they already use, with permissions inherited from the platform and every answer traceable to source transactions.
That is the launch. The launch is also, deliberately, the smaller story.
The bigger story: a foundation built for an AI moment that has not settled
With MCP, symmetRE is releasing something bigger than a feature. The connector gives clients access to a shared AI foundation the company has spent years building, designed for the one problem real estate firms run into every time they try to put AI to work on a real portfolio: the data underneath.
"Frontier models are incredibly powerful. The reason most CRE owners can't harness the full potential has nothing to do with the models," says Jack Swoboda, co-founder and CEO of symmetRE. "It has to do with the layer underneath. Real portfolio data is fragmented across every property management system your operators control, defined differently at every firm, and continuously moving as books backfill and post-close adjustments compound. That problem does not get easier when a new model ships. We have been working on solving it for years. The MCP connector is what makes that work directly reachable from whatever AI tool a client already uses."
The foundation underneath the connector is what symmetRE believes constitutes a durable AI edge for clients:
- Validated, not raw. What MCP exposes is the same data set that drives symmetRE's dashboards today, already cleaned, standardized, and computed using each firm's own definitions. Models reason over numbers symmetRE has already validated, not raw warehouse data with definitions scraped from the internet.
- Traceable, all the way down. Every answer can be traced through the platform to source transactions in the underlying property management systems (general ledger transactions, unit-level details, etc.). Not a citation stapled to a summary row.
- Permissioned by inheritance. The connector displays only what the signed-in user already has permission to see. No new access to govern; no new permissions to administer.
- A modular and extensible framework. symmetRE's AI capabilities (including an in-product chatbot, an AI Summary Widget for PDF reports, and now MCP) are composed from the same foundation: a tool library, a unified provider abstraction that lets any model plug in, and a context layer that is expanding to hold the meaning of each firm's formulas, variables, and operating methodology. The architectural decision is deliberate.
- Model-agnostic by design. Because the connector is built on MCP — an open protocol now supported across every major AI provider — clients are not locked to any single model vendor. The same question can be asked of Claude and ChatGPT side by side; clients can switch models when a better one ships; the work above the foundation does not need to be rebuilt as the model market evolves.
"We don't ask the model to do your math," says Artur Gaifutdinov, symmetRE's chief technology officer. "Your company's formulas run in our computation layer deterministically, and the model presents what's already been computed. Ask how recurring CapEx split from growth CapEx last quarter — you get your firm's split. Not an LLM's average of how the internet defines those categories."
What comes next: symmetRE as an AI partner, not an AI feature
The MCP connector is the first release on the foundation. It is not the last. Through the rest of the year, symmetRE is committed to a set of strategic directions that extend the platform from a place where AI answers questions to a place where AI creates with clients' data.
The categories of work the company is investing in:
- From AI that explains to AI that creates. The next set of AI capabilities authors with portfolio data rather than only describing it. Generating new charts, dashboards, and report templates on demand, all grounded in the same validated data and the same firm-specific definitions.
- AI that operates on each firm's methodology. Continued investment in the context layer (per-client knowledge bases, per-client formula context, and cross-client formula reference) so the AI consistently applies a client's math, their definitions, their operating logic rather than generic defaults.
- Continuity across conversations. Memory and persistence so the AI remembers what was asked before, what changed since, and how a decision played out over time.
- AI in the tools you already use. MCP is the first. Native integrations with Excel and connections to other AI tools your team relies on will follow, shaped by what our clients tell us is most useful.
"Most vendors added AI as a feature," Gaifutdinov said. "We're trying to be the AI partner for firms that want to use this seriously — which means doing the work that doesn't get easier when a new model ships. MCP is one capability on that foundation. There will be many more."
About symmetRE
symmetRE is the portfolio-wide reporting and intelligence platform for real estate owners and operators. The company builds the ingestion, semantic, and computation infrastructure that turns fragmented property management data into a single, validated data and AI foundation — making portfolio analytics, AI workflows, and operator reporting reliable at production scale and across asset classes. symmetRE is headquartered in Chicago and serves owners, operators, investors, and asset managers across the United States. Learn more at symmetRE.com.
Media Contact
Lindsay Curry
Head of Marketing, symmetRE
lindsay.curry@symmetre.com
SMM Plus Expands Telegram Engagement Testing Program for Telegram Creators
SMM Plus, a Telegram-focused growth and visibility platform, provides a testing-centric framework for channel owners, digital brands, and communities that want to evaluate engagement metrics before they launch large-scale promotional efforts. By offering testing services through the expanded program, SMM Plus aims to reduce uncertainty regarding audience quality, visibility performance, and Telegram social proof within the Telegram ecosystem. The expanded testing program offers support to Telegram creators who want a transparent way to assess performance indicators of Telegram channel growth and engagement visibility.
Why Telegram Channels Need Testing Before Scaling
While the volume of competition across Telegram communities is increasing, many channel owners are having a difficult time evaluating the authenticity of their audiences, as well as evaluating consistency in their engagement, and overall visibility performance.
The relevance of this issue is growing in the educational community, crypto-focused channels, media publishers, agencies, and other digital brands that utilize Telegram as a major communication platform.
Many creators are investing in their growth activities without fully understanding how the engagement signals they receive will impact their channel's perception, Telegram audience signals of trust, and visibility of the content being created.
Industry experts expect to see an increase in the use of Telegram social proof indicators (such as engagement with posts, keeping up with the consistency of members in the group, the pacing of engagement, etc.) as a means of evaluating Telegram communities in the future.
The testing-first approach of SMM Plus allows creators to utilize these indicators and helps reduce the risk of implementing a larger scale growth strategy based on those indicators.
Telegram Testing Tools Added to Expanded Initiative
As part of their newly expanded initiative, SMM Plus has launched a number of testing tools that will measure different types of growth indicators and the behavior of audience members within Telegram. As it currently stands, this testing will occur in the existing testing ecosystem through:
- Visibility of Telegram premium members
- Evaluation of Telegram post views
- Engagement of Telegram reactions
- Member retention monitoring
- Tracking of BotStart performance
Each type of testing will supply creators with the ability to see all the different forms in which audiences signal to them and how visible their engagement will be in the Telegram community.
Testing-First Telegram Growth Approach
The company has said that instead of focusing on numerical growth, they will evaluate how visibility indicators, along with engagement patterns among users of their channels, contribute to the trust and credibility of those channels.
The company’s stated goal of the initiative is to encourage creators on Telegram to use an analytical and performance-based approach for building their channels. The company also mentioned that many creators are now increasingly comparing the quality of their engagement rates and how consistent their retention rates are, as well as comparing user behaviour with regard to visibility of their channels before increasing their campaigns or working with a third-party growth provider.
“The Telegram community is evolving to be much more sophisticated in how they assess the level of Telegram channel trust and engagement they have with Creators," stated Michael Carter, Head of Growth Strategy at SMM Plus. "As a result, creators no longer just look at how many people are connected with their channels. They now review visibility signals, quality of interaction and how their channel will appear to new visitors before making long-term growth decisions.”
He also added that creators may benefit from having an environment that tests and validates Telegram channel growth and engagement with users to help them feel more confident about making long-term growth decisions.
As competition for audience attention continues to intensify, industry analysts have also pointed to a larger trend within the industry toward validating Telegram performance and promoting transparency of engagement signals between social media and messaging platforms.
Telegram Growth Trends and Creator Behavior
As more and more communities join Telegram, the visual authority and the engagement perception become crucial elements of their growth strategy. It has been suggested that creators begin to evaluate the effectiveness of their channels according to their evaluation metrics, engagement rate and Telegram performance indicators based on interactions within the Telegram community.
Furthermore, the growth through testing could also be attributed to shifting behavior of creators who rely heavily on their experiments and performance metrics when choosing campaigns and audiences.
SMM Plus announced that its initiative will adapt accordingly to changing preferences of creators as well as Telegram engagement trends.
About SMM Plus
SMM Plus is a technology company specializing in Telegram visibility, engagement analysis, and social performance ecosystems. The company develops products that facilitate the analysis of Telegram audience signals, interaction metrics, channel engagement indicators, and overall growth trends among creators on the platform. Its ecosystem includes visibility testing solutions, engagement analytics tools, and Telegram growth products designed to help users better understand and optimize their performance on Telegram. For more information, please visit smm.plus.
Media Contact
SMM Plus Media team
support@smm.plus
+44 7363 085699

FoundersX Ventures: SpaceX IPO to Catalyze $28 Trillion AI-Space Convergence and Orbital Data Center Era
FoundersX Ventures, a proud investor in SpaceX, today highlighted the transformative potential of SpaceX’s upcoming IPO as a major catalyst for the convergence of AI and space infrastructure — a market the company values at over $28.5 trillion.
In its recent S-1 filing, SpaceX outlined a bold vision where space becomes critical infrastructure for the next generation of AI. With Starship’s reusability driving launch costs down dramatically, the company is positioned to unlock orbital data centers, real-time space-based AI processing, and enterprise AI applications at global scale.
“SpaceX is not just revolutionizing access to space — it is becoming the foundational infrastructure provider for the AI age,” said Helen Liang, founder and managing partner of FoundersX Ventures. “We are incredibly excited about the upcoming IPO. By slashing launch costs from a historical average of $18,500 per kilogram down to $1,400 with SpaceX's Falcon Heavy, and eventually to below $500 per kilogram, SpaceX will make building large-scale data centers in orbit not only viable but economically compelling. This will trigger explosive growth in orbital compute, AI infrastructure, and a new wave of innovation across industries.”
FoundersX has strategically invested across the full stack of this emerging AI-Space ecosystem:
- SpaceX: The clear leader enabling low-cost access to space and global connectivity through Starlink.
- Turion Space: Developing agile DROID satellites for space domain awareness, satellite inspection, rendezvous operations, and future in-space servicing — critical infrastructure for a crowded and active orbital environment.
- FS2: Building next-generation, high power-efficiency chips using 2D materials to address the AI compute memory wall, enabling more efficient AI training and inference both on Earth and in orbit.
- World Labs: Building advanced spatial intelligence and generative 3D world models. Its technology enables highly realistic simulation of complex orbital environments, making it possible to virtually design and optimize large-scale data centers in space before physical deployment.
- Charge Robotics: Automating solar farm construction to rapidly scale renewable energy capacity needed to power the surging demand of AI data centers. The company could take on building solar farms in space.
- mBodi AI: Creating an intuitive robotics brain that allows industrial robots to learn complex tasks through natural language and demonstrations, accelerating physical automation for manufacturing and infrastructure. The company could take on robotics training for building data centers in space.
As AI demand continues to surge, the need for massive, power-hungry data centers is outstripping Earth’s available land, energy, and cooling capacity. Orbital data centers offer compelling advantages: unlimited solar power, natural cooling in space, and low-latency global connectivity via Starlink.
FoundersX believes SpaceX’s ability to slash launch costs by nearly 90% will be the tipping point that makes orbital AI infrastructure a reality, creating enormous opportunities across compute, energy, robotics, and satellite technologies.
“We backed SpaceX 6 years ago because we saw its potential to fundamentally reshape multiple industries,” added Liang. “The upcoming IPO marks a pivotal moment — not just for SpaceX, but for the entire ecosystem that will build the future of AI in space.”
About FoundersX Ventures
FoundersX Ventures is a Silicon Valley-based venture capital firm focused on investing in pioneering tech companies building AI infrastructure and disruptive solutions across industries. The firm partners with visionary founders building transformative solutions to shape the future. Beside Silicon Valley, the firm has offices in Boston and New York City. Selected investments: SpaceX, World Labs, Turion Space, Universal Quantum, Charge Robotics, FS2, Salt Security, Jeeves, Kapital, Alinea Invest, and Solve Intelligence.
Media Contact
Tor Parawell
tor.parawell@foundersx.com

World's First Global Energy Drink Index Finds European Products Outperform North American Rivals on Quality
Pat Eckert, a certified water sommelier and internationally recognised independent beverage professional based in Meckesheim, Germany, assessed products from all six inhabited continents using a 36-criteria framework covering measurable product quality, ingredients, transparency and formulation standards. Top-performing products were submitted for independent laboratory testing and analytical verification.
The project, called the Six Continents Index, is believed to be the world's first objective global ranking of the category, revealing that energy drinks sold in North America differ so fundamentally from their counterparts elsewhere that they are, in effect, a different product. Six Continents Index was designed to be professional, rigorous and free of commercial influence.
What began as an attempt to identify the world's best energy drink became something considerably broader. The data revealed that the category does not follow a single global product philosophy. Regional differences in pasteurisation rates, sugar use, artificial sweetener reliance and vitamin content are so pronounced that the same product category effectively describes different things depending on the continent.
Key Findings
Among the most significant findings from the index:
- 85.7% of European energy drinks were pasteurised, compared with 12% in North America and under 1% in South America.
- 78.9% of energy drinks in Asia used real sugar. The equivalent figure for North America was 8%.
- 84% of North American energy drinks relied entirely on artificial sweeteners, against 4.2% in Europe and near zero across Asia, Australia, South America and Africa.
- Australian products averaged 4.2 vitamins per unit, compared with 2.9 in North America.
- Aspartame, classified by the World Health Organization's International Agency for Research on Cancer as "possibly carcinogenic to humans" (Group 2B), was detected in 10.5% of products assessed globally. Of those, 43% came from Africa.
- BPA-free labelling was found on just 1.4% of products in the global sample.
- North America — the world's largest energy drink market by revenue — ranked last among the six continents in the overall index.
Rankings
At the continental level, Europe achieved the highest overall score. Australia and Oceania ranked second, followed by Asia in third place.
At the brand level, HELL Energy of Hungary achieved the highest overall score for objective product quality across the entire index. Second place went to 28 BLACK of Germany, followed by TAKE OFF, also from Germany.
Notable Observations
Two brands stood out for reasons beyond the rankings. Red Bull was the only energy drink label identified in virtually every market assessed worldwide. Japan's Lipovitan-D was the oldest brand in the study, having been on the market since 1962.
"Nobody had ever applied a consistent global framework to this category before. What we found was not just that some products are better than others — it was that the category itself means different things in different parts of the world," said Pat Eckert, lead researcher of the Six Continents Index.
Methodology
Assessed brands were not notified in advance, did not apply to participate and had no involvement in the evaluation. No paid participation, sponsorship or commercial influence played any role in the project. Full methodology and further findings are available on request.
About Six Continents Index
The Six Continents Index was led by Pat Eckert and his team at Fine Liquids, Meckesheim, Germany. Eckert is a German certified water sommelier and independent beverage expert whose previous work has been featured by The Guardian, ABC News, The Telegraph, L'Express, Der Spiegel and the BBC. For more information, visit sixcontinentsindex.com.
Disclaimer
The Six Continents Index is an independent research project conducted by Fine Liquids and led by Pat Eckert. Rankings and findings are based on the study's methodology, selected product samples, and data available during the research period and are intended for informational purposes only. References to health or regulatory classifications are provided for context and do not constitute medical advice or safety determinations regarding any specific product. Product formulations may vary by market and change over time. Claims that the index is the "world's first" are based on the researchers' review of publicly available information and are qualified accordingly.
Media Contact
Pat Eckert
pat@fine-liquids.com

GoodVision AI Introduces the '7-Layer AI Cake' Framework for the Inference Era
GoodVision AI has introduced what it calls the “7-Layer AI Cake” framework, outlining how the company believes the AI industry will evolve as global infrastructure shifts from a model-centric era toward a token-driven economy.
According to the company, the future AI stack will consist of seven interconnected infrastructure layers:
- Power infrastructure
- AIDC (AI data centers)
- GPUs
- Large language models (LLMs)
- Token distribution networks
- Intelligent scheduling systems
- AI agents
GoodVision AI argues that AI is evolving into a large-scale “token industrial system,” where the ability to generate, distribute, orchestrate, and optimize tokens efficiently may become more important than model size alone.
The company said the framework was developed in response to the rapid expansion of AI inference workloads, as AI agents become increasingly embedded across enterprise workflows, consumer applications, robotics, edge devices, and autonomous systems.
Over the past two years, the AI industry has largely been defined by the race to build increasingly powerful foundation models. Parameter counts expanded from hundreds of billions to trillions, while GPU clusters scaled from thousands of chips to tens of thousands. The industry focused heavily on model capability, reasoning performance, and the pursuit of AGI.
But according to GoodVision AI, the next phase of AI may be driven less by model scale itself and more by the infrastructure required to support global token generation, routing, and consumption at scale.
Layer 1: Energy Emerges as the Foundation of AI Infrastructure
As AI infrastructure expands globally, energy is becoming one of the industry’s most important bottlenecks. Large AI data centers can now consume as much electricity as mid-sized cities, while power grid expansion in many regions is struggling to keep pace with surging AI demand.
According to GoodVision AI, this imbalance is pushing the AI industry further upstream into energy infrastructure. The company believes stable baseload energy sources, long-term power access, and energy efficiency will become increasingly strategic as inference workloads continue scaling globally. In the emerging token economy, access to reliable, low-cost energy may become one of the most important competitive advantages in AI infrastructure.
Layer 2: AI Data Centers Become “Token Factories”
The company’s framework positions AI data centers, or AIDCs, as the “token factories” of the AI era. Rather than relying on individual GPUs, modern AI infrastructure depends on large-scale GPU clustering capable of producing tokens at industrial scale.
While traditional AI data center development cycles can take years, power grid expansion often takes even longer. As AI inference demand accelerates, many legacy infrastructure models are struggling to keep pace. At the same time, AI infrastructure is gradually shifting away from purely hyperscale centralized architectures toward more distributed and modular systems.
According to GoodVision AI, inference workloads are increasingly expected to move closer to end users through regional edge-oriented deployment models. Against this backdrop, the company is pursuing a modular AI Factory strategy centered around lightweight, rapidly deployable inference infrastructure. Rather than relying solely on hyperscale facilities, GoodVision AI says it is focusing on smaller inference-oriented AI Factory nodes designed for dense regional deployment and faster integration with local energy infrastructure. The company argues that modular AI Factories are better aligned with the long-term evolution of distributed inference networks.
Layer 3: GPUs Become the Production Equipment of the Token Economy
If electricity represents the energy foundation of AI, GPUs represent the production equipment. During the first phase of the AI boom, GPU demand was driven primarily by model training. But the next wave of infrastructure growth is increasingly expected to come from inference. Unlike training workloads, which remain concentrated among a small number of frontier AI companies, inference workloads are expected to expand across nearly every application, device, and endpoint. Robotics, AI wearables, autonomous systems, and future AI agent collaboration networks all require continuous real-time inference — and therefore continuous token generation and consumption.
According to industry observers, the future of AI infrastructure may ultimately depend on one key metric: how efficiently tokens can be generated per unit of time. As a result, infrastructure layers surrounding GPUs — including networking, power management, liquid cooling, servers, and optical interconnects — are becoming increasingly critical to AI performance and operational efficiency. Industry analysts increasingly view this infrastructure layer as the “picks-and-shovels” foundation of the broader AI economy.
Layer 4: LLMs Evolve Into “Token Production Engines”
Large language models are also evolving beyond simple demonstrations of model capability.
According to GoodVision AI, the market is increasingly shifting away from a pure race for parameter scale and toward a broader focus on inference efficiency, deployment cost, orchestration capability, and scalability. The company argues that models themselves do not directly create value. Instead, value emerges through continuous inference — the repeated generation, routing, and consumption of tokens across real-world applications.
As a result, LLMs are increasingly becoming the “token production engines” of the AI economy. Competition at the model layer is also evolving rapidly. Rather than focusing solely on parameter counts, the market is increasingly evaluating models based on factors such as inference efficiency, token generation cost, long-context processing, multi-agent collaboration, and integration with distributed infrastructure systems.
According to GoodVision AI, future winners at the model layer may not simply be those building the largest models, but those capable of operating models efficiently at global scale. The company says it is developing its own optimization strategy by deploying large language models directly within its AI Factory infrastructure, allowing it to evolve from a traditional compute rental provider into a Token-as-a-Service platform.
Layer 5: Token Distribution Emerges as the “Power Grid” of the AI Era
As AI infrastructure scales globally, another major challenge is becoming increasingly important: how compute resources can be distributed and utilized efficiently at scale. Token distribution networks may ultimately function much like the electrical grids of the industrial era — connecting fragmented GPU resources into unified global infrastructure systems.
As AI adoption expands beyond frontier model developers into enterprises, startups, applications, and AI agent ecosystems, the demand for flexible and scalable compute distribution is rising rapidly. At the same time, the market is beginning to shift away from purely centralized cloud architectures toward more distributed compute networks optimized specifically for inference workloads.
Lighter deployment models, faster provisioning systems, lower-latency routing, and cost-efficient GPU access are becoming increasingly important as inference workloads continue scaling globally.
According to GoodVision AI, token distribution infrastructure is emerging as one of the key connective layers of the AI economy — linking GPUs, AI models, edge nodes, and inference demand into a scalable global compute network.
Layer 6: Intelligent Scheduling Could Become the “Brain” of the AI Economy
As AI infrastructure becomes more distributed, intelligent scheduling and token orchestration are emerging as critical infrastructure layers for the next phase of AI. The future challenge is no longer simply whether sufficient compute exists, but whether compute can be utilized intelligently and efficiently.
The company argues that not every workload should be routed to the most expensive frontier models. Lightweight tasks may be processed locally, privacy-sensitive workloads may remain at the edge, and high-concurrency inference may increasingly rely on hybrid orchestration systems. As a result, AI infrastructure is evolving toward dynamic scheduling architectures capable of routing workloads across different models, compute environments, and inference layers in real time.
GoodVision AI compares this infrastructure layer to modern intelligent power grid systems, where multiple energy sources can operate simultaneously while efficiency depends on dynamic orchestration systems operating in the middle layer. The company believes future AI architectures will increasingly rely on a simple principle: “the right model running on the right compute for the right task.”
According to GoodVision AI, this transition could fundamentally reshape the economics of AI infrastructure, shifting the industry away from simply “selling compute” toward “optimizing compute” through orchestration, inference routing, and token scheduling systems.
Layer 7: AI Agents Could Become the Largest Consumers of Tokens
The final layer of the framework focuses on AI agents. According to GoodVision AI, AI agents may ultimately become the largest drivers of global token consumption.
Unlike traditional AI applications, AI agents can simultaneously call multiple models, tools, APIs, and inference systems while continuously performing reasoning, coordination, planning, and autonomous execution. As a result, future token consumption could far exceed the scale of today’s human-AI interactions.
The company believes the future AI economy may involve not only billions of humans using AI systems, but potentially tens or even hundreds of billions of AI agents continuously operating and interacting with one another. At that scale, the primary bottleneck may shift away from model capability itself and toward token orchestration efficiency.
According to the company, AI agents are gradually evolving from simple software applications into continuously operating economic participants within a global intelligent infrastructure network.
The Next Phase of AI May Depend on Fully Integrated Infrastructure
Despite rapid growth across the AI sector, GoodVision AI believes the industry remains structurally fragmented. Some organizations possess advanced GPU infrastructure but remain constrained by energy supply. Others operate large-scale AI data centers but lack efficient orchestration systems. Some have developed powerful AI models and agents but continue to face high inference costs and latency bottlenecks.
According to the company, the next phase of AI competition will center on connecting these fragmented infrastructure layers into a unified global system.
The company argues that the future AI economy will no longer revolve solely around training increasingly large models. Instead, billions of continuously operating AI agents will require coordinated systems spanning energy, compute, networking, orchestration, and distributed inference infrastructure.
Industry observers increasingly view this transition as a shift away from software-centric infrastructure toward a much broader industrial system spanning semiconductors, cloud computing, energy systems, networking, and intelligent orchestration.
According to GoodVision AI, the most important AI companies of the future may not necessarily be those with the single largest models, but those capable of integrating energy, compute, networking, models, and token flows into one scalable infrastructure system.
About GoodVision AI
GoodVision AI is an AI infrastructure company led by former AWS and IBM executives, building intelligent compute scheduling and distributed edge inference systems for the next generation of AI applications. Founded in 2019, the company’s platform helps enterprises address challenges around token consumption, latency, and cost as AI workloads scale. GoodVision AI combines centralized cloud infrastructure, localized edge compute, and proprietary orchestration technology to dynamically route workloads based on complexity, latency sensitivity, and cost. With infrastructure expansion across Asia and the U.S., the company is building a globally distributed compute network to support real-time, large-scale AI inference.
Media Contact
Joy Chen
media@goodvision.ai